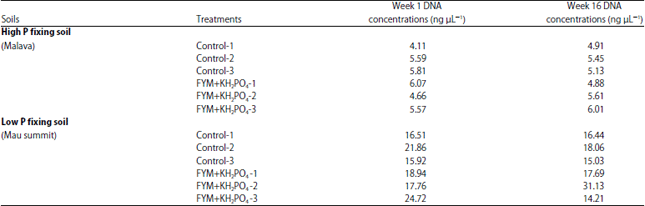
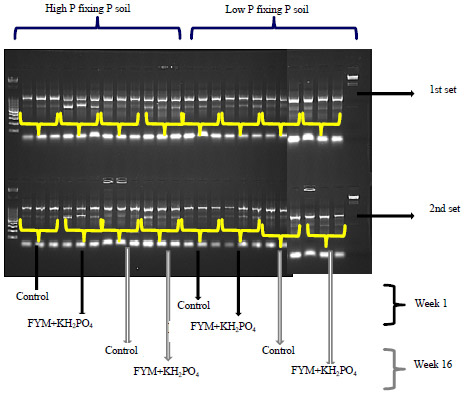
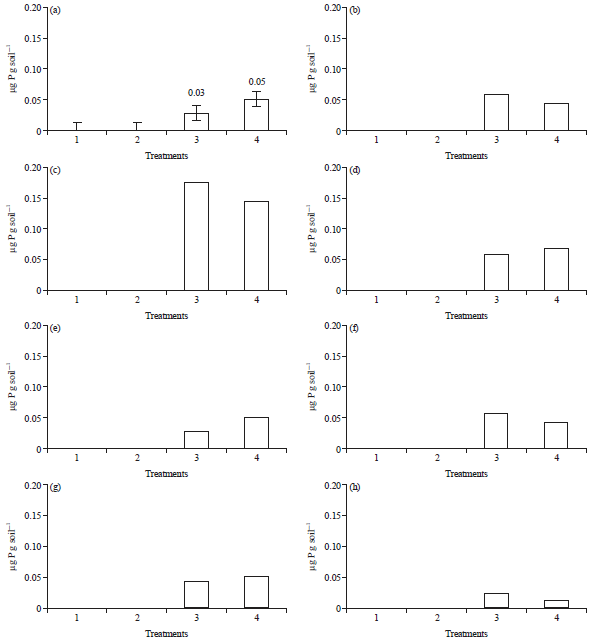Research Article
Changes in Phosphorus Turnover and Microbial Communities of Two Kenyan Soils
Centre de Recherche sur la Durabilite et la Productivite des Plantations Industrielles (CRDPI), B.P. 1291, Pointe-Noire, Republic of the Congo
LiveDNA: 682.13136
George Ayaga
Kenya Agricultural and Livestock Research Organization, KALRO, P.O. Box 278, Busia, Kenya
Philip Brookes
Department of Soil Science, Rothamsted Research, Harpenden, Herts. AL5 2JQ, United Kingdom