ABSTRACT
When conducting incubation experiments to measure soil respiration in response to a given treatment in soils of different texture, it is important to have baseline information about the soils and to ensure that the effect of the storage or pre-incubation conditions on the results are minimized. Three experiments were conducted with up to seven soils (3 non-saline, 4 saline) to investigate (1) The relationship between soil water content or water potential and soil respiration, (2) The time required until soil respiration is stabilized after rewetting of air-dry soil and (3) If pre-incubation at a given matric or osmotic potential changes the response of soil respiration to matric or osmotic potential. The texture of the soils varied from sand to sandy loam. Cumulative respiration showed an optimum curve in response to differential soil water content in all 7 soils, but the maximal cumulative respiration was reached at different water content in the soil, being 45 g kg–1 for the non-saline sand and 200 g kg–1 for the non-saline sandy loam. However, when expressed against water potential, maximal cumulative respiration was achieved between 0 and -1 MPa in all soils. Rewetting of air-dry soil induced a flush of respiration in the first 1-2 days, but respiration rates stabilized 7-8 days after rewetting in all soils. Pre-incubation at low matric or osmotic potential for two weeks did not significantly affect soil respiration at low matric or osmotic potential in the following two weeks compared to soils which had been pre-incubated at optimal matric potential and high osmotic potential. In conclusion, when comparing the response of respiration to water availability among soils of different texture it is important to consider water potential and not water content. Furthermore, an adaptation period of 2 weeks does not seem to increase the tolerance of soil respiration to low osmotic or matric potential compared to non-adapted soils.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijss.2015.117.129
URL: https://scialert.net/abstract/?doi=ijss.2015.117.129
INTRODUCTION
Microbial activity is strongly affected by soil water content, increasing with increasing soil water content from dry to moist soil but then decreasing again (Harris, 1981; Killham, 1994). The increase is due to greater water and nutrient availability as the pores become increasingly water-filled. Beyond the optimal soil water content however, further increases in water content decrease microbial activity due to limited gas exchange and thus anaerobic conditions.
After collection, soils are often air-dried and then sieved to remove large particles and stones. The soils are then stored air-dry before being used for experiments. Microbial activity is very low in air-dry soil due to lack of water and nutrients. Many microorganisms survive by becoming dormant or by forming spores. Therefore, air-drying also minimizes carbon (C) loss during storage. In Mediterranean climate such as that in Southern Australia, top soils often dry during early summer and remain air-dry for several months. Hence, air-drying and storage of air-dry soil mimics the conditions in the field.
Rewetting of air-dry soil causes a flush of respiration which is due to the release of previously accumulated osmolytes in microbial cells and of organic matter within aggregates (Kaiser et al., 2015; Kieft et al., 1987; Halverson et al., 2000; Denef et al., 2001; Fierer and Schimel, 2003). This flush occurs within a few minutes after rewetting, with a peak in the first day, but then decreases rapidly due to consumption of the labile C (Kieft et al., 1987; Fierer and Schimel, 2003). The size of the flush depends on the C content of the soil and also the size of the microbial biomass being greater in soils with high C content and/or microbial biomass C (Butterly et al., 2009, 2010). Respiration rates comparable to those in moist soil are often reported to be found 5-10 days after rewetting (Kieft et al., 1987; Fierer and Schimel, 2003; Mikha et al., 2005). Compared to continuously moist soil, rewetting also causes a change in microbial community structure, particularly in soils with higher C availability (Butterly et al., 2009, 2010). This flush in respiration after rewetting of air-dry soil may mask treatment effects that are to be investigated in an experiment. Therefore, it is important to rewet the soils before imposing any treatments to allow microbial activity to return to equilibrium. The time until this equilibrium is reached may vary with soil type. Hence, it is important to test this for any soil to be used.
Water availability is not only a function of the matric potential but also, particularly in saline soils, of osmotic potential. Low osmotic potential, caused by high salt concentrations in the soil solution draws water out of the cells and thus inhibits microbial activity. Whereas, sensitive cells are killed at low osmotic potential, tolerant microorganisms accumulate osmolytes (Oren, 2001; Killham and Firestone, 1984; Schimel et al., 1989; Beales, 2004).
Microbial communities may be more tolerant to a stressor such as low osmotic or matric potential, if they are given time to adapt. During this adaptation period, sensitive genotypes die, whereas tolerant genotypes survive and become dominant. In previous studies, microbial activity recovered within two days of exposure to high heavy metal concentrations (Diaz-Ravina and Baath, 1996) or a pH change (Pettersson and Baath, 2004), indicating that a new, more tolerant community has developed. However, it is not known how quickly microbial communities adapt to low matric or osmotic potential.
In the field, microbial communities are able to adapt to low osmotic or low matric potential, because they are usually exposed to these stressors for weeks, months or even years. Hence, sensitivity to a low water potential may be overestimated in experiments, where microbial communities are suddenly exposed to the stressor and not given time to adapt. Therefore, it needs to be investigated, if pre-incubation of a soil at low osmotic or matric potential makes the microbial communities more resistant to these stresses than, if the stress is imposed suddenly.
The aims of the experiments described in this study were to: (1) Determine the soil water content that results in maximal cumulative respiration for the soils, (2) Determine the time until respiration reached constant rates after rewetting of air-dry soil in the soils and (3) Assess the effect of adaptation on sensitivity to different matric and osmotic potential.
MATERIALS AND METHODS
Seven soils were used, three non-saline and four saline soils. A non-saline sand, a non-saline sandy loam and the four saline sandy loam soils were collected from Monarto, South Australia (35°05'S and 139°06'E). Another non-saline sandy loam was collected from Mount Bold, South Australia (38.11°S 138.69°E). The particle size analysis of each soil was determined by hydrometer method (Day, 1965). The EC and pH were measured in a 1:5 soil: Water suspension after 30 min settlement following 1 h end-over-end shaking at 25°C.
| Table 1: | Properties of the soils used in the experiments |
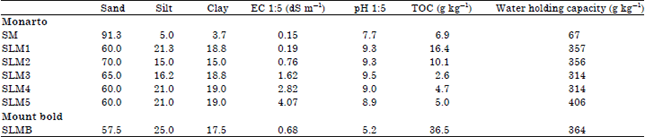 | |
| S: Sand, SL: Sandy loam, TOC: Total organic carbon and EC: Electrical conductivity | |
Total Organic Carbon (TOC) was determined by dichromate oxidation described by Walkley and Black (1934). Water Holding Capacity (WHC) was determined using suction and pressure techniques (Klute, 1986) at -0.01 MPa. Each soil was placed in cores of 1 cm diameter and 0.5 cm height in 4 replicates using the appropriate amount of air-dry soil to maintain the bulk density according to their texture. Soils were thoroughly wetted with NaCl solutions according to their EC. Separate pressure plates were used for different EC’s. Soils were allowed to drain for 7 days at -0.01 MPa pressure in pressure chambers. Oven dry weight of soil was determined after drying at 105°C for 24 h. The properties of the soils are given in Table 1.
Determination of the water retention curves: The water retention curves of the soils were determined using suction and pressure techniques (Klute, 1986). Matric potential was estimated from the moisture retention curve using the following equation (Hillel, 1980):
where, Ψ is water potential, a, b is empirical constants, θ is water content.
For the four saline soils, the water retention curves were determined by equilibrating the soils with saline solutions according to their EC. The saline solutions of the appropriate EC were prepared with NaCl salt using the following equation (Robert Murray, personal communication):
where, y is NaCl concentration, g L–1 and x is EC of solution, dS m–1.
The electrical conductivity was determined in a 1:5 soil: Water mixture (EC1:5) after 1 h end-over-end shaking. The EC1:5 was converted to the EC of a saturated paste (ECe) using the following equation (Rengasamy, 2006):
The osmotic potential was estimated using the following equation of Richards (1954):
where, Ψπ is the soil osmotic potential (MPa) at the actual moisture content, θact of the soil and Ecmeas is the measured electrical conductivity (dS m–1) of an extract with a water content θref (= 5 g g–1 for a 1:5 soil: water mixture).
| Table 2: | Water content and corresponding matric potential in the seven soils in experiment 1 |
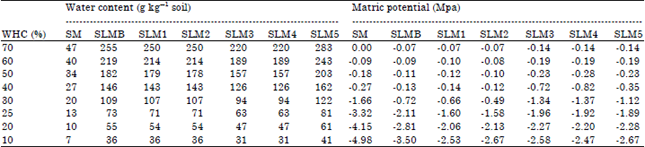 | |
| S: Sand, SL: Sandy loam, TOC: Total organic carbon and WHC: Water holding capacity | |
Respiration measurements: The soils (25 g, oven dry basis) were added to PVC cores (diameter 3.7 cm, height 5 cm) with a nylon mesh base (0.75 μm, Australian Filter Specialist) and packed to a bulk density (http://www.pedosphere.com/resources/bulkdensity/triangle_us.cfm) according to their texture: 1.55 g cm–3 (sand) and 1.46 g cm–3 (sandy loam). The cores were placed immediately into 1 L glass incubation jars and sealed with gas tight lids equipped with septa to allow headspace sampling. The glass jars were incubated in the dark at 22-25°C. Respiration was quantified by measuring headspace carbon dioxide (CO2) concentrations every 24 h using a Servomex 1450 infra-red gas analyser (Servomex Group, Crowborough, England). After each measurement, the jars were opened to equilibrate the CO2 to ambient concentrations and then resealed. The CO2 concentrations were measured immediately after resealing the jars. The CO2 evolved from each sample was calculated as the difference between the initial (after resealing of the jars) and the CO2 concentrations after 24 h.
Experiment 1: The aim of experiment 1 was to determine the soil water content that results in maximal cumulative respiration for the soils.
All seven soils mentioned above were used. The soils were incubated moist for 10 days. This incubation time was chosen based on previous experiments conducted in our lab which had shown that respiration rates stabilized 7-10 days after rewetting of air-dry soil in a range of different soils. The water content was based on a subjective assessment: The soils were not too dry and not too wet. Following this pre-incubation, the soils were dried by spreading them in a large glass dish and placing the dish in fan forced oven at 25°C for up to three hours until they had reached the desired water content. Then, pea (Pisum sativum L.) straw (C/N 26, water soluble C 27 g kg–1), ground and sieved (0.25-2 mm), was mixed into the soils (2% w/w) to provide a readily-available nutrient source. Respiration was measured until the respiration rates remained stable for at least 2 days which was between 5 and 10 days after addition of the pea straw. There were 2 replicates per treatment. The water content and corresponding matric potential is shown in Table 2.
Experiment 2: The aim of experiment 2 was to determine the time until respiration reached constant rates after rewetting of air-dry soil in the soils to be used in the subsequent experiments.
All seven soils mentioned above were used. The soils were rewet to the water content that resulted in maximal respiration (determined in Experiment 1) and respiration measured for 18 days. By then respiration rates had remained constant for at least 10 days. There were 2 replicates per treatment.
Experiment 3: The aim of experiment 3 was to assess the effect of adaptation on sensitivity to different matric and osmotic potential. The non-saline sandy loam was used for this experiment. The general treatment structure is shown in Table 3.
| Table 3: | Design for experiment 3 |
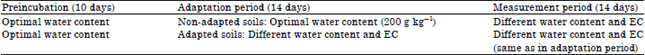 | |
| EC: Electrical conductivity | |
| Table 4: | ECe and water content in treatments of experiment 3 with corresponding osmotic and matric potential |
 | |
| ECe: Electrical conductivity (saturated phase) | |
Two sets of soils were exposed to different conditions in the adaptation period (14 days): (1) Non-adapted: Incubated at optimal soil water content and (2) Adapted: Incubated at different water content and EC. After this adaptation period, the non-adapted soils were adjusted to the same water content and EC as the adapted soils while the water content and EC of the adapted soils was maintained. Pea straw (2% w/w) was added and respiration measured over 14 days (measurement period).
The EC levels used were ECe 0.7, 6.0, 7.7, 17.3, 34.3, 42.0 dS cm–1 (referred as EC0.7, EC6.0, EC7.7, EC17.3, EC34.4 and EC42.0) which were achieved by adding 0, 1.5, 2, 5, 10 and 12.5 g NaCl kg–1 soil. The soil water content was maintained constant at 200 g kg–1 which is optimal for this soil.
The soil water content was adjusted to 90, 100, 120, 130, 150 and 200 g kg–1 (referred to as WC90, WC100, WC120, WC130 and WC200) as described for experiment 1. The matric and osmotic potentials in the different treatments are shown in Table 4.
There were 3 replicates per treatment. Significant differences between different treatments in respiration rate and cumulative respiration were assessed by 2-way ANOVA (adaptation×EC or water content) and Tukey test with α = 0.05 (GenStat® for Windows 8.0, VSN Int. Ltd, UK, 2005).
RESULTS
The moisture retention curves are presented in Fig. 1 for 5 soils; the other sandy loam soils from Monarto had similar retention curves to those shown. Whereas, the five sandy loams have quite similar retention curves, the soil water content of the sand decreased more rapidly, reaching very low matric potentials at higher water contents than in the sandy loam soils.
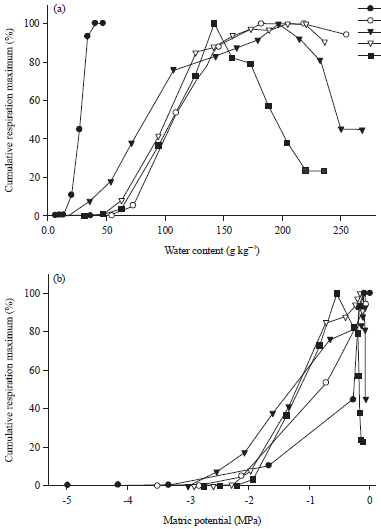
Experiment 1: Cumulative respiration was affected by water content. In the sandy loam soils, cumulative respiration increased with increasing water content initially, but then decreased (Fig. 2a, for 5 soils, the other sandy loam soils had similar retention curves to those shown).
In the sand, there was no decrease in cumulative respiration at the higher water contents. The response of cumulative respiration to different water content varied between soils with some being more affected than others. The water content that resulted in maximal respiration was much lower in the sand than in the sandy loam soils. And among the sandy loam soils, it was lowest in SLM4.
The water contents that resulted in maximal cumulative respiration was 45 g kg–1 for the non-saline sand and 200 g kg–1 for the non-saline sandy loam (Fig. 2). In the five sandy loam soils it ranged from 140-240 g kg–1. These water contents are referred to as optimal water content in this thesis.
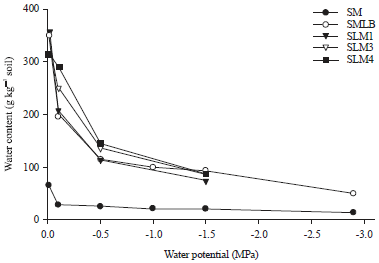 | |
| Fig. 1: | Water retention curves of a sand and four sandy loam soils |
 | |
| Fig. 2(a-b): | Cumulative respiration in percentage of maximum in a sand and four sandy loam soils as a function of (a) Water content and (b) Matric potential |
However, when cumulative respiration is plotted against matric potential, the response is very similar between the five soils (Fig. 2b).
 | |
| Fig. 3(a-b): | Respiration rates in the seven soils over 38 days after rewetting |
| Table 5: | Parameters for logarithmic equations of water retention curves of the soils. Matric potential (MPa) is inserted into the equation in positive values. Water content is in g 100 g–1 soil |
 | |
| r2: Water content | |
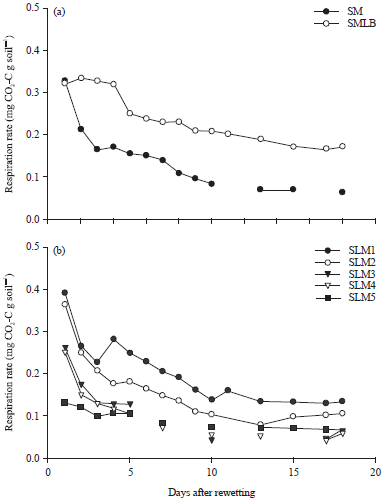
Experiment 2: Respiration rates were high on the first day after rewetting but then declined rapidly in all seven soils (Fig. 3). The respiration rates stabilized after 7-8 days and then remained constant until day 18 when the experiment was stopped.
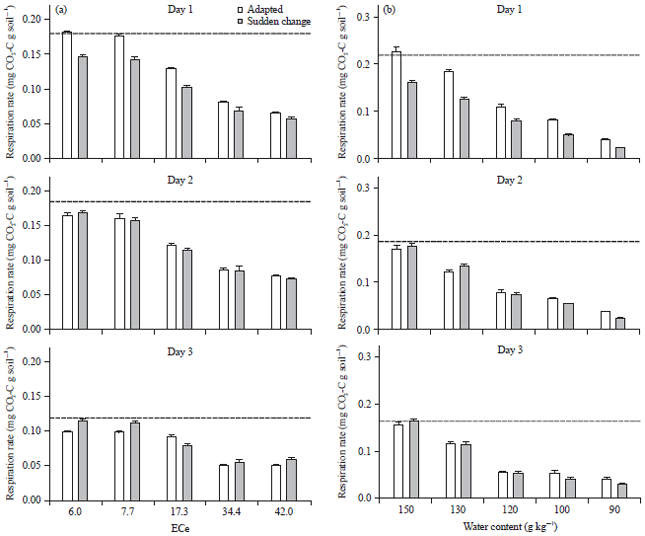
Experiment 3: The matric potential ranged between -0.10 and -1.46 MPa while the osmotic potential ranged between -0.05 and -3.21 MPa (Table 5). Respiration rates and cumulative respiration were strongly affected by EC and water content, both decreasing with increasing EC and decreasing water content (Fig. 4-5).
For example, respiration rate on day 1 compared the non-saline soils was less than 30% at EC42.0 and compared to the optimal soil water content, 20% at WC90 (Fig. 4).
 | |
| Fig. 4(a-b): | Respiration rates in the first 3 days after residue addition and for the non-adapted soils, the changes in (a) Osmotic and (b) Matric potential. Dashed line indicated respiration rates of the control (optimal water content, no salt addition) |
In contrast to the strong effect of EC and water content on respiration, the effect of adaptation on respiration rate was small and transient. Significant differences in respiration rate between adapted and non-adapted soils were detected only during the first 3 days. On day 1, respiration rates were higher in adapted compared to non-adapted soils with generally greater differences at lower EC and higher water content. Compared to day 1, respiration rates on day 2 were lower in adapted soils but remained similar in non-adapted soils. There were no significant differences in respiration rates between adapted and non-adapted soils on day 2. The same is true for the respiration rates on day 3 in the soils incubated at different water content. In the soils with different ECs, respiration rates on day 3 were significantly higher in non-adapted soils than in adapted soils up to EC 7.7 but not at higher EC levels. The decrease in respiration rates from 2-3 days was greater in adapted than in non-adapted soils.
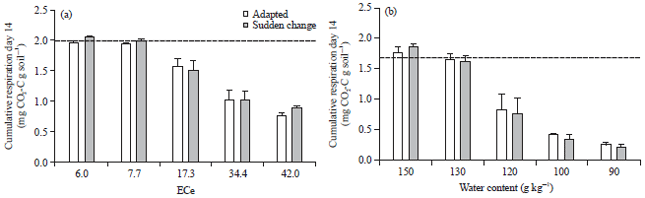
Cumulative respiration after 14 days was similar in adapted and non-adapted soils and strongly affected by EC and water content (Fig. 5). Cumulative respiration compared to the non-saline soils was 40% at EC42.0 and compared to the optimal soil water content 13% at WC90.
 | |
| Fig. 5(a-b): | Cumulative respiration 14 days after residue addition and for the non-adapted soils, the change in (a) Osmotic and (b) Matric potential. Dashed lines indicates respiration rates of the control (optimal water content, no salt addition) |
DISCUSSION
The strong response of soil respiration to soil water content (Fig. 2) is in agreement with many previous studies (Harris, 1981; Killham, 1994) and can be explained by the changes in water and oxygen (O2) availability. In dry soil, water films around aggregates are very thin and water is held tightly on to aggregate surfaces resulting in a lack of water for metabolic activity and diffusion of substrates to the microbes limits their activity (West et al., 1989; Ilstedt et al., 2000). As the water content increases, the thickness of the water film around aggregates increases and small pores become water-filled. The water content optimal for microbial activity is reached when the soil contains enough water to allow diffusion of substrates to the microbes and enzymes away from them, but large pores are still air-filled to allow gas exchange, i.e. removal of CO2 from respiration and supply of O2 since most soil microorganisms are aerobic (Ilstedt et al., 2000). If the water content increases further, gas exchange is impaired because diffusion of gases is 103-104 times lower in water than in air (Armstrong, 1979). Hence, anaerobic conditions develop, limiting the activity of aerobe microorganisms. Differential anaerobic microbial communities develop using a succession of alternative electron acceptors from nitrate at high redox potential over manganese and iron to low molecular weight compounds, such as acetic acid at very low redox potential from which methane is produced (Marschner, 1995; Liesack et al., 2000).
The differential response of the different soils to changes in water content is related to their texture. Coarse textured soils have few small pores in which water is retained but many large pores to allow gas exchange. Therefore water and not oxygen availability governs microbial activity and higher water contents are required for optimal activity in coarse textured soils. Fine textured soils on the other hand, have many small pores to retain water, but very few large pores for gas exchange. Thus, in fine textured soils, oxygen availability limits microbial activity and lower water contents are required for optimal activity.
Due to these differences in water retention properties, it is important to determine the water content that results in optimal microbial activity for each soil to ensure maximal activity in the control soils and/or during pre-incubation before the start of an experiment. Moreover, it is not the water content per sec but the availability of the water to microorganisms that determines their activity; water availability is best reflected in water potential, because it is a measure of how much energy is required to remove water from the soil. Therefore, in order to compare the effect of water availability on microbial activity in soils, it is necessary to relate microbial activity to water potential not water content (Fig. 2b). This is particularly important when comparing different soils since the matric potential at a given soil water content varies between soils (Fig. 1). Thus, in order to expose microbes in different soils to the same water stress (matric potential), they have to be adjusted to different water contents, higher water content in coarse soils, lower in fine textured soils (Fig. 2b).
Respiration was also strongly affected by EC (osmotic potential) in the sandy loam (Fig. 4-5) showing that not only matric potential, but also osmotic potential affects microbial activity. The negative effect of salinity on microbial activity is in agreement with many other studies (Pathak and Rao, 1998; Rietz and Haynes, 2003; Yuan et al., 2007), but in most of these studies, only the EC is given. However, the EC is a poor measure of salinity stress to microbes, because it does not necessarily reflect the salt concentration in the soil solution. At the same EC1:5 or ECe, the salt concentration in the soil solution is most likely higher in a coarse textured soil than in a fine textured soil because the former has a lower water content. Thus the same amount of salt (measured as EC) is contained in a smaller amount of water, increasing its concentration and thus decreasing the osmotic potential.
The flush in respiration after rewetting of air-dry soil followed by a rapid decrease within 2-4 days (Fig. 3) is in agreement with many previous studies (Kieft et al., 1987; Fierer and Schimel, 2002; Xiang et al., 2008; Butterly et al., 2009) and can be explained by the release of easily decomposable organic compounds upon rewetting and their depletion. The stabilisation of the respiration rates after about 8 days after rewetting is also in accordance with previous studies (Butterly et al., 2010). These steady-state respiration rates may be somewhat higher than in soils that have been stored moist, because they contain more C which would have been decomposed during storage in moist soils. Additionally, the community composition of the air-dried and rewet soils may differ from those in moist soils (Hamer et al., 2007; Butterly et al., 2009). Nevertheless, it can be assumed that the microbial community in the air-dried and rewet soils is established and in equilibrium within 8 days after rewetting. Thus, any effect of a treatment imposed after this period can be attributed to the treatment itself and is not confounded by the previous drying and rewetting.
It was hypothesized that a sudden decrease in matric or osmotic potential may not allow soil microbes to adapt and therefore, have a more detrimental effect than in soils, where the microbes had time to adapt. Surprisingly, cumulative respiration of adapted and non-adapted soils was the same after the 14 day experimental period (Fig. 5). Thus, prior exposure to low matric or osmotic potential for 14 days did not result in greater tolerance to low matric and osmotic potential compared to soils, where the potential was adjusted just prior to the start of the respiration measurements. However, there were some small and transient differences in the first 3 days after adding the residues (Fig. 4). On the first day, after addition of the residues, respiration rates were higher in the adapted soils compared to the non-adapted soils which had just experienced a change in water potential. However, on day 2, after addition of residues, the respiration rates in the adapted soils were lower than on day 1 whereas, they remained stable in the non-adapted soils. On day 3, the respiration rates declined in adapted and non-adapted soils with a smaller decrease in the non-adapted soils. Thus, the adapted microbes could respond more quickly to the increased substrate availability than the microbes which had were exposed to a change in water potential immediately before residue addition. However, the slower decrease in respiration rates in the first three days in the non-adapted soils suggests that a tolerant community developed quickly and could utilise the substrates, whereas, the easily decomposable compounds had already been decomposed in the adapted soils. Thus, in the adapted soils, the increase in respiration was rapid but short-lived whereas, it was delayed but more sustained in the non-adapted soils. This differential time course explains, why there were no differences in cumulative respiration between adapted and non-adapted soils after 14 days.
Therefore, the hypothesis that a sudden decrease in matric or osmotic potential may not allow soil microbes to adapt, have to decline and therefore have a more detrimental effect than in soils where the microbes had time to adapt. The results suggests, that although a sudden change in water potential transiently reduces microbial activity, microbes can adapt to decreases in water potential within a few days and then utilise available substrates to the same extent as adapted microbes.
However, it should be noted that due to the lack of easily available substrates, microbial activity was very low during the adaptation period in all treatments, therefore, it is likely that only a small proportion of the microbial community was active and those that were active in the adapted soils may not have been able to accumulate osmolytes. After addition of the pea straw, many of the previous inactive microbes would become active. These freshly activated microbes in the adapted soils may therefore experience a sudden stress and the need to accumulate osmolytes similar to the microbes in the non-adapted soils. Therefore, to ascertain if adaptation increases tolerance to low water potential, easily available substrates should be added during the adaptation period to allow a greater proportion of the microbial population to be active and hence, adapt.
CONCLUSION
These experiments provided the following information that is important for studies with soil salinity:
| • | For the seven soils to be used, the water retention curve was determined which allows adjusting the soils to a desired matric potential. This will enable comparison between soils of different texture, because the basis for this comparison will be matric potential, hence, the actual work needed to withdraw water from the soil and not water content |
| • | The determination of the water content (matric potential) that results in maximal activity for each soil is important to ensure that matric potential is not limiting microbial activity during the pre-incubation and in the control treatments |
| • | The constant respiration rates 8 days after rewetting of air-dry soil show that a per-incubation period of 10 days is sufficient to achieve a stable microbial activity; it can be assumed that the microbial community has recovered from the effect of storage and rewetting |
| • | Since, adapted and non-adapted microbial communities do not seem to differ substantially in their response to low water potential, differential water potentials can be imposed at the start of the experimental period in experiments |
REFERENCES
- Beales, N., 2004. Adaptation of microorganisms to cold temperatures, weak acid preservatives, low pH and osmotic stress: A review. Compr. Rev. Food Sci. F., 3: 1-20.
CrossRefDirect Link - Butterly, C.R., E.K. Bunemann, A.M. McNeill, J.A. Baldock and P. Marschner, 2009. Carbon pulses but not phosphorus pulses are related to decreases in microbial biomass during repeated drying and rewetting of soils. Soil Biol. Biochem., 41: 1406-1416.
CrossRefDirect Link - Butterly, C.R., P. Marschner, A.M. McNeill and J.A. Baldock, 2010. Rewetting CO2 pulses in Australian agricultural soils and the influence of soil properties. Biol. Fert. Soils, 46: 739-753.
CrossRefDirect Link - Day, P.R., 1965. Particle Fractionation and Particle Size Analysis. In: Methods of Soil Analysis, Black, C.A., D.D. Evans, L.E. Ensminger, J.L. White and F.E. Clark (Eds.). American Society of Agronomy, Madison, WI., USA., pp: 545-567.
Direct Link - Denef, K., J. Six, H. Bossuyt, S.D. Frey, E.T. Elliott, R. Merckx and K. Paustian, 2001. Influence of dry-wet cycles on the interrelationship between aggregate, particulate organic matter and microbial community dynamics. Soil Biol. Biochem., 33: 1599-1611.
CrossRefDirect Link - Diaz-Ravina, D. and E. Baath, 1996. Development of metal tolerance in soil bacterial communities exposed to experimentally increased metal levels. Applied Environ. Microb., 62: 2970-2977.
Direct Link - Fierer, N. and J.P. Schimel, 2002. Effects of drying-rewetting frequency on soil carbon and nitrogen transformations. Soil Biol. Biochem., 34: 777-787.
CrossRefDirect Link - Fierer, N. and J.P. Schimel, 2003. A proposed mechanism for the pulse in carbon dioxide production commonly observed following the rapid rewetting of a dry soil. Soil. Sci. Soc. Am. J., 67: 798-805.
Direct Link - Halverson, L.J., T.M. Jones and M.K. Firestone, 2000. Release of intracellular solutes by four soil bacteria exposed to dilution stress. Soil. Sci. Soc. Am. J., 64: 1630-1637.
CrossRefDirect Link - Hamer, U., M. Unger and F. Makeschin, 2007. Impact of air-drying and rewetting on PLFA profiles of soil microbial communities. J. Soil Sci. Plant Nutr., 170: 259-264.
CrossRefDirect Link - Ilstedt, U., A. Nordgren and A. Malmer, 2000. Optimum soil water for soil respiration before and after amendment with glucose in humid tropical acrisols and a boreal mor layer. Soil. Biol. Biochem., 32: 1594-1599.
CrossRefDirect Link - Kaiser, M., M. Kleber and A.A. Berhe, 2015. How air-drying and rewetting modify soil organic matter characteristics: An assessment to improve data interpretation and inference. Soil Biol. Biochem., 80: 324-340.
Direct Link - Kieft, T.L., E. Soroker and M.K. Firestone, 1987. Microbial biomass response to a rapid increase in water potential when dry soil is wetted. Soil Biol. Biochem., 19: 119-126.
CrossRefDirect Link - Killham, K. and M.K. Firestone, 1984. Salt stress control of intracellular solutes in Streptomycetes indigenous to saline soils. Applied Environ. Microb., 47: 301-306.
Direct Link - Liesack, W., S. Schnell and N.P. Revsbech, 2000. Microbiology of flooded rice paddies. FEMS Microbiol. Rev., 24: 625-645.
CrossRefDirect Link - Marschner, H., 1995. Mineral Nutrition of Higher Plants. 2nd Edn., Academic Press Ltd., London, New York, ISBN-13: 978-0124735439, Pages: 889.
Direct Link - Mikha, M.M., C.W. Rice and G.A. Milliken, 2005. Carbon and nitrogen mineralization as affected by drying and wetting cycles. Soil Biol. Biochem., 37: 339-347.
CrossRefDirect Link - Oren, A., 2001. The bioenergetic basis for the decrease in metabolic diversity at increasing salt concentrations: Implications for the functioning of salt lake ecosystems. Hydrobiologia, 466: 61-72.
CrossRefDirect Link - Pathak, H. and D.L.N. Rao, 1998. Carbon and nitrogen mineralization from added organic matter in saline and alkali soils. Soil Biol. Biochem., 30: 695-702.
CrossRefDirect Link - Pettersson, M. and E. Baath, 2004. Effects of the properties of the bacterial community on pH adaptation during recolonisation of a humus soil. Soil. Biol. Biochem., 36: 1383-1388.
CrossRefDirect Link - Richards, L.A., 1954. Diagnosis and improvement of saline and alkali soils. Soil. Sci., 78: 7-33.
Direct Link - Rietz, D.N. and R.J. Haynes, 2003. Effects of irrigation-induced salinity and sodicity on soil microbial activity. Soil Biol. Biochem., 35: 845-854.
CrossRefDirect Link - Schimel, J.P., W.J. Scott and K. Killham, 1989. Changes in cytoplasmic carbon and nitrogen pools in a soil bacterium and a fungus in response to salt stress. Applied Environ. Microb., 55: 1635-1637.
Direct Link - Walkley, A. and I.A. Black, 1934. An examination of the degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci., 37: 29-38.
CrossRefDirect Link - West, A.W., G.P. Sparling and T.W. Speir, 1989. Microbial activity in gradually dried or rewetted soils as governed by water and substrate availability. Aust. J. Soil. Res., 27: 747-757.
CrossRefDirect Link - Xiang, S.R., A. Doyle, P.A. Holden and J.P. Schimel, 2008. Drying and rewetting effects on C and N mineralization and microbial activity in surface and subsurface California grassland soils. Soil. Biol. Biochem., 40: 2281-2289.
CrossRefDirect Link - Yuan, B.C., Z.Z. Li, H. Liu, M. Gao and Y.Y. Zhang, 2007. Microbial biomass and activity in salt affected soils under arid conditions. Applied Soil. Ecol., 35: 319-328.
CrossRefDirect Link








