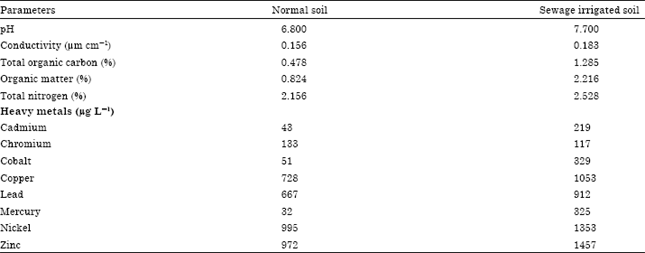
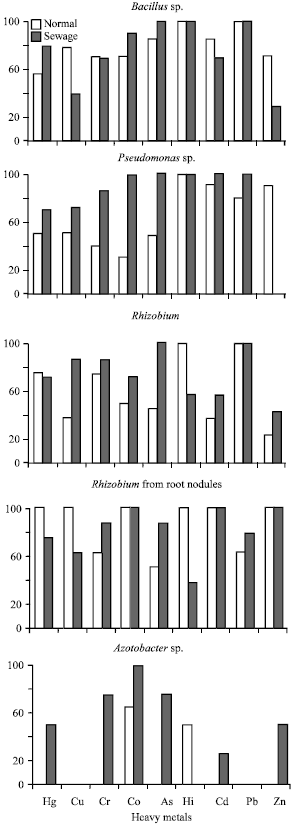
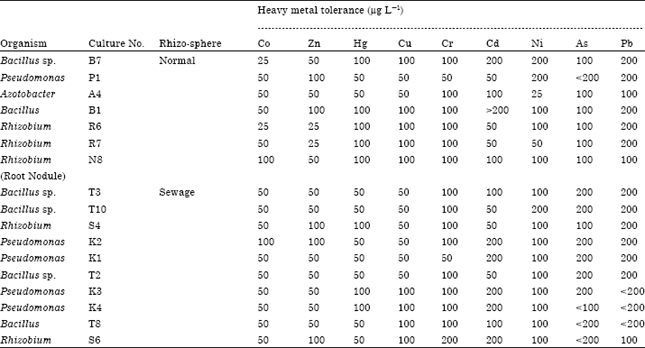
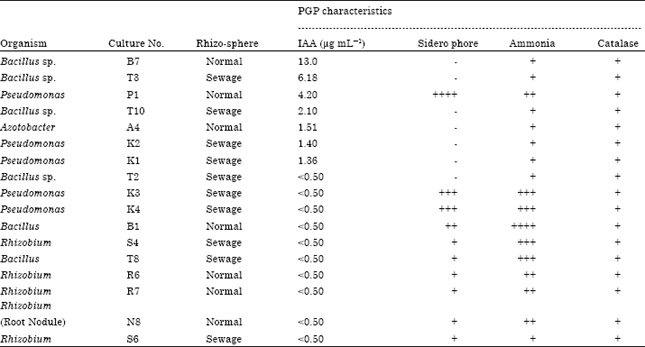
Research Article
Pisum sativum and Associated Plant Growth Promoting Rhizobacteria: Effect of Normal and Sewage Irrigation
Department of Biological Sciences, School of Basic Sciences, Sam Higginbottom Institute of Agriculture, Technology and Sciences, Allahabad 211 007 Uttar Pradesh, India
Babu Joseph
College of Applied medical Sciences, P.B. No. 1383, Shaqra University, Shaqra11961, Kingdom of Saudi Arabia
Abin Mani
Directorate of Research, Sam Higginbottom Institute of Agriculture, Technology and Sciences, Allahabad 211 007 Uttar Pradesh, India
Sonia Chacko
Directorate of Research, Sam Higginbottom Institute of Agriculture, Technology and Sciences, Allahabad 211 007 Uttar Pradesh, India