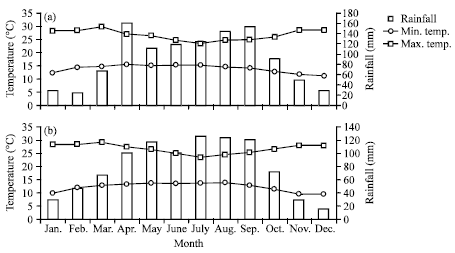
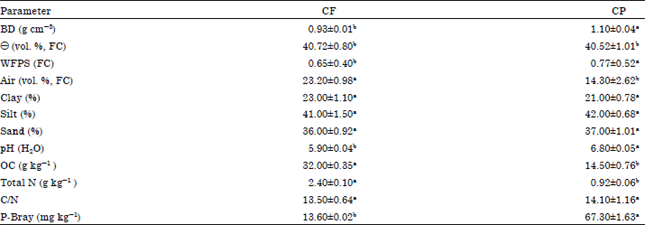
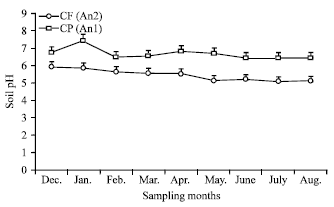
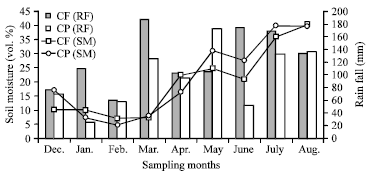
Research Article
Nitrogen Mineralization Dynamics under Different Moisture Regimes in Tropical Soils
Department of Plant and Environmental Sciences, Norwegian University of Life Sciences, P.O. Box 5003, N-1432 Aas, Norwa
Endalkachew Wolde-meskel
School of Plant and Horticultural Sciences, Hawassa University, P.O. Box 05, Hawassa, Ethiopia
Sheleme Beyene
School of Plant and Horticultural Sciences, Hawassa University, P.O. Box 05, Hawassa, Ethiopia
Lars R. Bakken
Department of Plant and Environmental Sciences, Norwegian University of Life Sciences, P.O. Box 5003, N-1432 Aas, Norwa