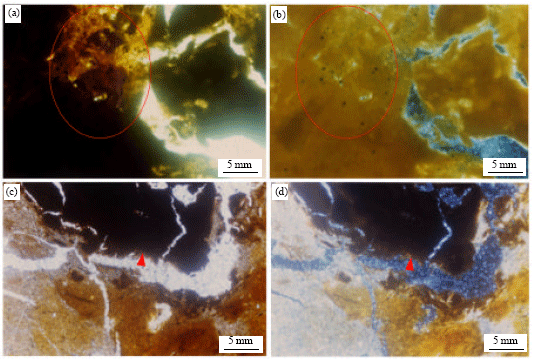
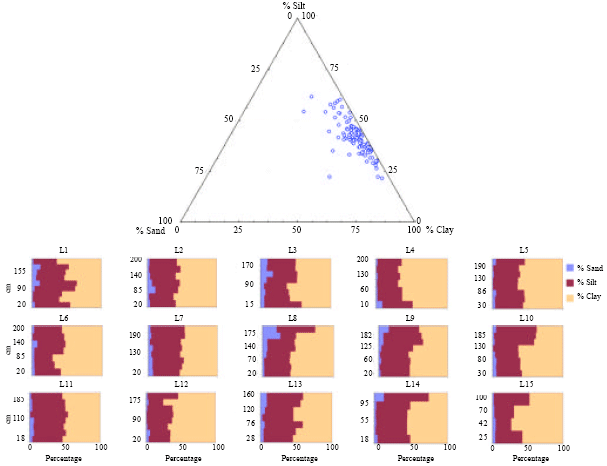
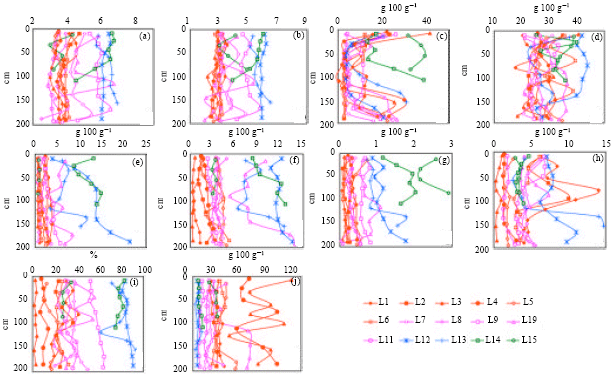
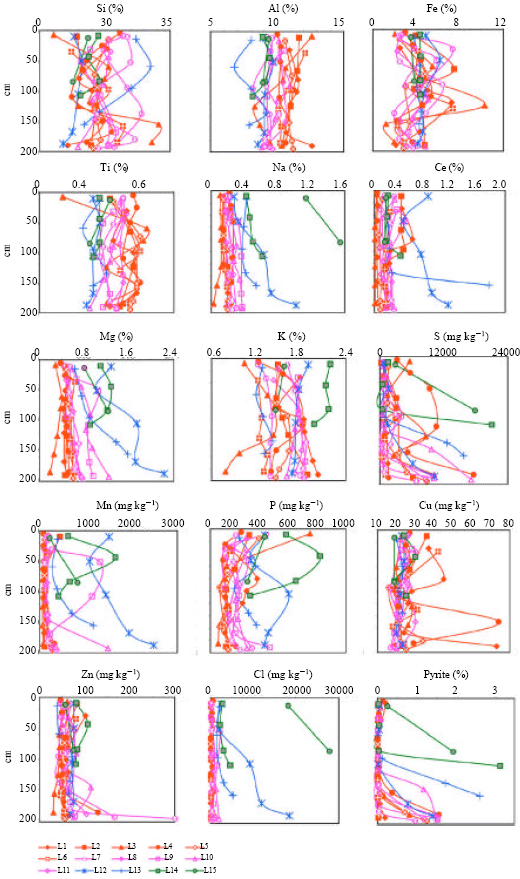
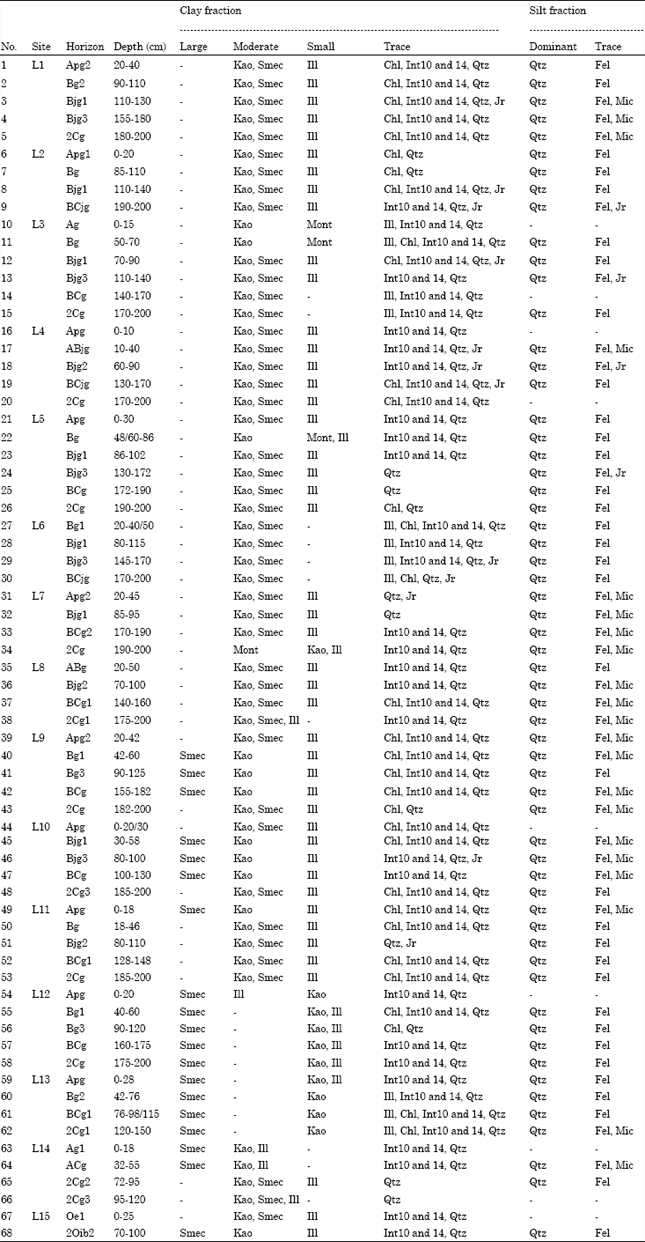
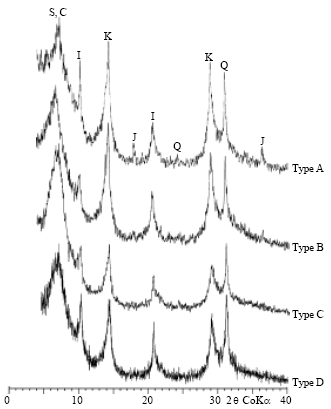
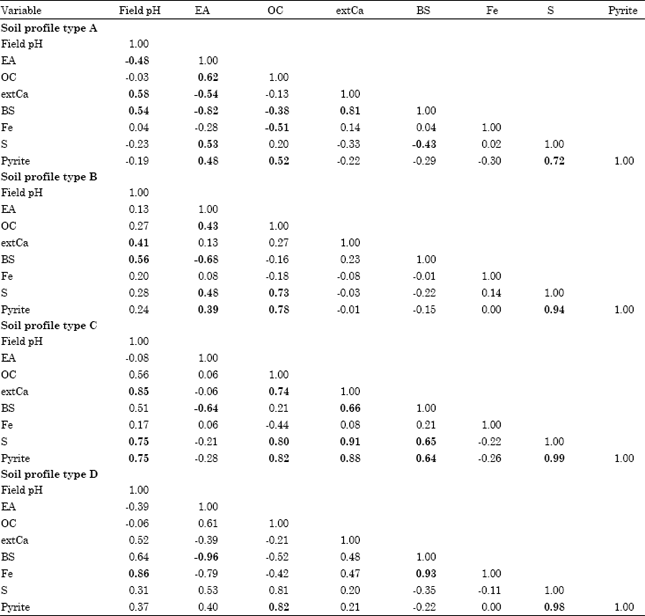
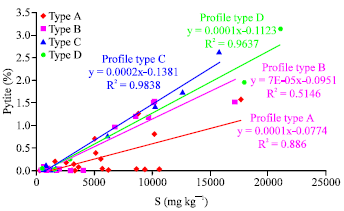
Research Article
Pedogenesis of Acid Sulfate Soils in the Lower Central Plain of Thailand
Office of Soil Resources Survey and Research, Department of Land Development, Chatuchak, Bangkok 10900, Thailand
M. Umitsu
Department of Geography, Nara University, Misasagi-cho, Nara 631-8502, Japan
S. Tawornpruek
Department of Soil Science, Faculty of Agriculture, Kasetsart University, Bangkok 10900, Thailand