Research Article
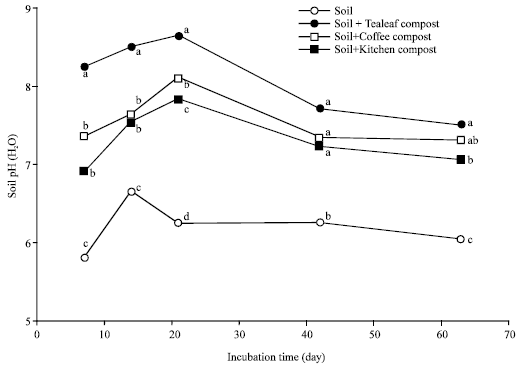
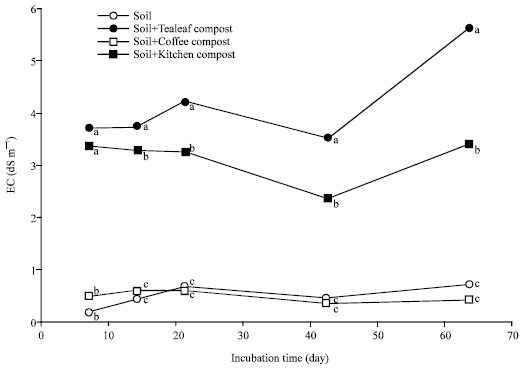
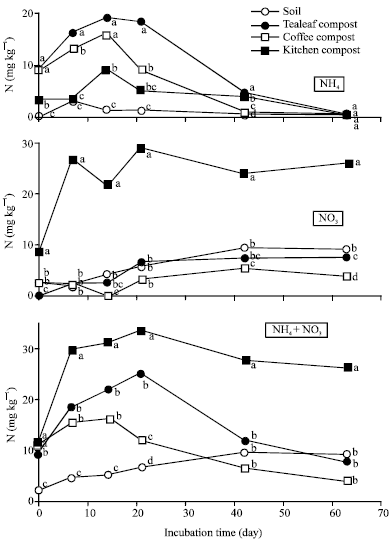
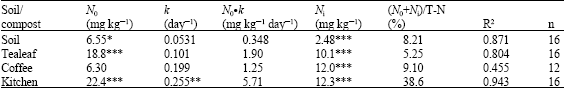
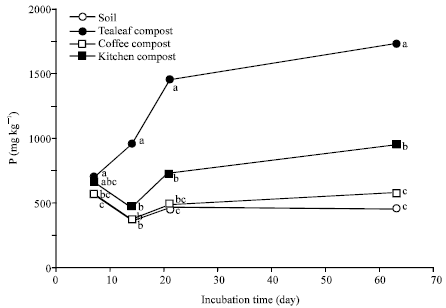
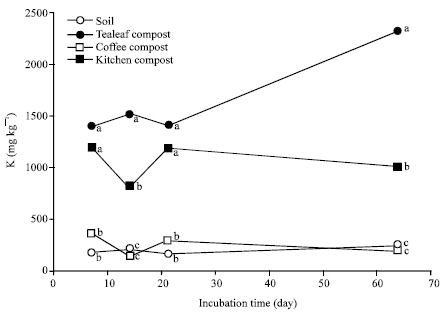
Nitrogen Mineralization Kinetics and Nutrient Availability in Soil Amended with Composted Tea Leaves, Coffee Waste and Kitchen Garbage
University Farm, Faculty of Agriculture, Ehime University, Hattanji, Ko 498, Matsuyama, Ehime 799-2424, Japan
Hideto Ueno
University Farm, Faculty of Agriculture, Ehime University, Hattanji, Ko 498, Matsuyama, Ehime 799-2424, Japan
Adel Ghoneim
University Farm, Faculty of Agriculture, Ehime University, Hattanji, Ko 498, Matsuyama, Ehime 799-2424, Japan