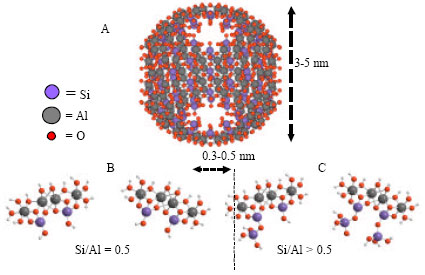
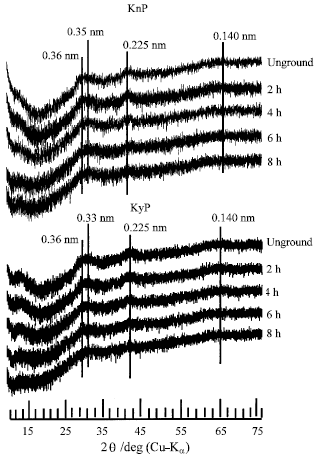
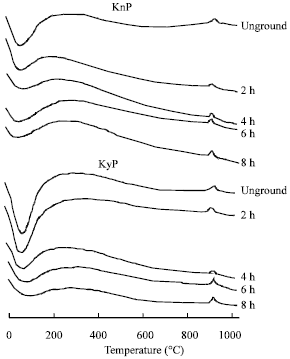
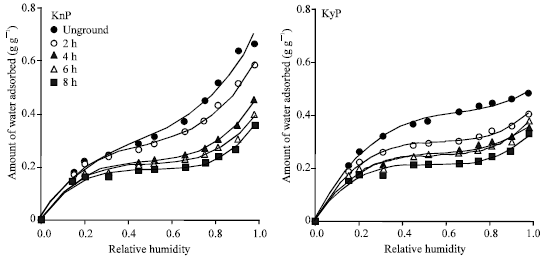
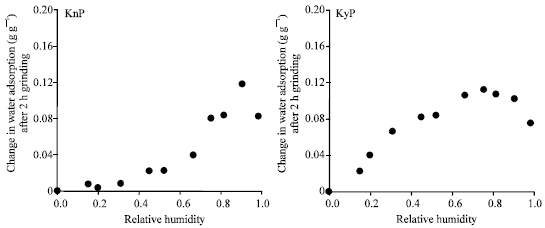
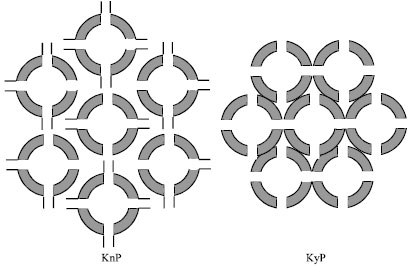
Research Article
Adsorption of Water on Nano-Ball Allophane as Affected by Dry Grinding
Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama 790-8566, Japan
Naoto Matsue
Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama 790-8566, Japan
Teruo Henmi
Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama 790-8566, Japan