Research Article
Basicity of Major Soil Groups in Lowland States of Southeastern Nigeria
Department of Soil Science and Technology, Federal University of Technology, P.M.B. 1526, Oweni, Nigeria
Mineral weathering is a natural source of cations that may be potentially adsorbed as exchangeable cations. Some of these cations are basic and include calcium (Ca) magnesium (Mg), potassium (K) and sodium (Na) and when combined are used to determine base saturation of soils. Perhaps, the greatest general influence of base saturation level is its effect on the availability of plant nutrients. In addition, basic cations are known to influence soils water content (Logsdon and Laird, 2004) and electrical properties (Nadler, 1999; Logsdon, 2000). Base saturation dominated by Ca and Mg showed significant (p<0.10) correlation with foliage values (Bailey et al., 2004). Horsley et al. (2000) reported that unhealthy stands of sugar maple were those with both low base cation nutrient concentrations and history of more severe insect defoliation. The exchangeable cation levels in the upper and lower B horizons are therefore useful predictors of stands plant health (Bailey et al., 2004), especially shrubby and tree crops.
In soils of southeastern Nigeria, soil erosion by water had devastated the landscape (Igwe, 2003) resulting in imbalances in the Ca: Mg and (Ca+Mg)/(Al+H) ratios (Oti, 2002). Osemwota et al. (2003) reported that Ca: Mg ratio could be a limiting factor in magnesium nutrition of maize crop. However, the origin, composition and distribution of these and other basic cations may vary with parent materials, irrespective of homogeneity in climate and land use patterns. Soils originating from glacier in Allegheny Plateau had higher base saturation than soils derived from unglaciated sites (Bailey et al., 2004). Alasiri (1994) reported that Ca:Mg interaction may not result in antagonism in soils derived from sedimentary rocks of southern Nigeria.
In Nigeria, Lal and Okigbo (1990) summarized causes of land degradation as bush fires deforestation, increasing intensity of cultivation, tillage-related practices, low input agriculture, accelerated erosion and construction works. These activities promote leaching of basic cations leaving a preponderance of acidic cations (Akamigbo, 1993; Igwe, 2003; Esu, 2005). But these pedogenic losses may differ with soil type. Based on this, the major objective of this study was to investigate the basicity of soil derived from different lithologies. It is hypothesized that the composition and distribution of basic cations differ with soil group.
Study Area
Abia and Imo States in Southeastern Nigeria constitute the study site. The site lies between latitudes 4°401 and 8°151N and longitudes 6°401 and 8°151 E. Soils are formed over 6 major parent materials, namely Alluvium, Coastal Plain Sands, Shale, Lower Coal Measures, Upper Coal Measures and Falsebedded Sandstone. It has a humid tropical climate, having among other features 9 months of wetness and 3 dry months. The study area has a rainforest vegetation. Subsistence agriculture is the main socio-economic activity. Multiple cropping is popular and are either cassava-based or yam-based. Land clearing is by slash and burn system and soil fertility maintenance is chiefly by bush fallowing. However, the fallow length (s) drastically shortened due (t) increasing population.
Field Work
The geology of the area guided the choice of sampling points in 2004. Six parent materials as earlier indicated were alluvium coastal plain sands, shale, lower coal measures and falsebedded sandstones and on each 5 profile pits were dug on its soils, giving a total of 30 profile pits for the purpose of this investigation. Soil samples were collected from the bottommost genetic horizon upwards to avoid contamination after soil profile pit description according to the procedure of FAO (1998). The collected soil samples were air dried, crushed and screened using 2 mm sieve to remove particles greater than 2 mm.
Laboratory Studies
Exchangeable cations (Ca, Mg, K and Na) were determined in 1 M NH4Cl extracts (Blume et al., 1990). Exchangeable aluminium (Al) and acidity were determined by titration (Thomas, 1982). Concentrations of cations in soil extracts were measured with direct-coupled plasma spectrophotometer. Effective Cation Exchange Capacity (ECEC) was determined as the sum of exchangeable Ca, Mg, K and Na and exchangeable acidity. The saturation value of exchangeable basic and acidic cations was obtained by dividing by ECEC expressed as a percentage (Cronan and Grigal, 1995). Particle size distribution was measured using the hydrometer method (Gee and Or, 2002).
Statistical Analysis
Soil data were subjected to analysis of variance (ANOVA) statistic using the PROC Mix-model of SAS (Little et al., 1996). Means were separated using standard error of the difference (SED) at 5% level probability.
Particle Size Distribution
Soils derived from upper coal measures and alluvium were very sandy when compared with other soil groups while soils formed over shale and lower coal measures were clayey (Table 1). There were significant (p≤0.05) differences in the particle size distribution resulting from variability in parent material (Table 1).
| Table 1: | Particle size distribution of soil in the study site (n = 150) |
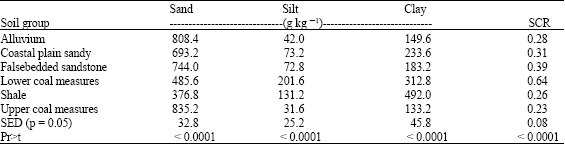 | |
| SED = Standard Error of the Difference, SCR = Silt Clay Ratio | |
| Table 2: | Variability in exchangeable cations (cmol kg -1) among soil groups |
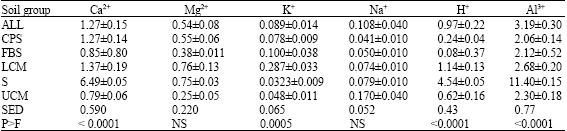 | |
| ALL = Alluvium, CPS = Coastal Plain Sands, FBS = Falsebedded Sandstones, LCM = Lower Coal Measures, S = Shale, UCM = Upper Coal Measures | |
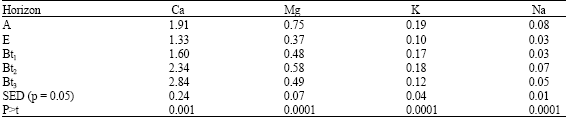
| Table 3: | Distribution of exchangeable basic cations (cmol kg-1) with horizon in the study area |
 | |
Exchangeable Cations
There were significant (p<0.0001) variations in the distribution of Ca2+, K+, H+ and Al3+ among soil groups (Table 2). Exchangeable Ca was highest in soils formed over shale and least in soil derived from upper coal measures. These variations are attributable to the composition of the materials from which soils are formed. Soils that originated from shale had the highest value of Ca since shale contains coarse sandstones with intercalations of thin shelly limestone materials. This is unlike soils from upper coal measures which consists of fragments of nodular ironstones plus ferruginized indurated shale and sandstones. Similar trend in Ca-distribution was generally followed by K+, H+ and Al3+ but Na+ was highest in Alluvial-derived soil, which can be due to marine deposits. It was also revealed that Shale-derived soils had high values of H+ and Al3+ despite the high Ca-content, suggesting possible influence of climate and land use history in these soils. High rainfall amount, duration and intensity may have increased leaching of basic cations (Oti, 2007), even in clayey soil of the agro-ecology.
Generally, basic cations decreased with depth except in Ca (Table 3). Although it was not investigated high surficial organic matter could be possible for this distribution while leaching may be responsible for Ca-increase with depth. Remarkably, least values of these basic cations were found in the horizon immediately below the epipedon, indicating that eluviations of basic cations possibly took place resulting to their illuviation in the argillic (Bt) horizons. The significant (p = 0.05) differences in the distribution of basic cations with depth is an indication of presence and activity of forces of pedogenesis in the soilsphere of these soil groups. While low K content portends poor K-nutrition by plants, low Na implies absence or low incidence of salinity problems in the study area. Argillic horizons in highly weathered topical soils like soil of he study site (SCR = 0.23-0.64) fix K leached and eluviated from surficial epipedons (Foth, 1984).
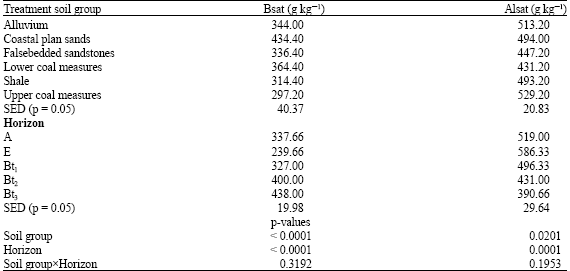
| Table 4: | Base-Aluminium saturations among soil groups and horizons |
 | |
| Table 5: | Elemental ratios |
 | |
Although, lost K can be recycled by roots of tree crops, it may not sufficiently supply the optimum needs of plant. Potassium availability in low exchangeable K soil as these depends on intensity, capacity and renewal rate (Wang et al., 2004). Very low levels of exchangeable Na suggestive of non-mobilization of some soil heavy metals such as cadmium and their reduced phytoavailability (Khoshgoftar et al., 2004).
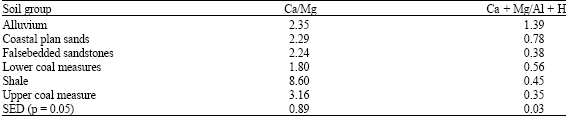
Base and aluminium saturations differed among soil groups and horizons (Table 4), with soils derived from upper coal measures and alluvium, indicating higher values of aluminium saturation (Alsat), having 529.20 and 513.00 g kg-1, respectively. Base saturations ranged from 297.2 to 434.4 g kg-1 in line with Oti (2007), who reported similar values in severely eroded soils of the agroecology. Among horizons highest concentration of Alsat were in E-horizon (586.33 g kg-1) and A-horizon (519 g kg-1), implying that these outermost soil horizons have suffered highest intensity of degradative forces of soil erosion, leaching and other redogenic processes of loss with minimal pedogenic additions. Highest basicity among soil groups and horizons was found in soil formed over coastal plain sands and Bt3-horizons. One would have expected soils formed over shale and lower coal measures to display very high basiciaty based on the mineralogy of these parent materials but the reverse was the case, meaning that other factors, such as climate, land use, land use history, age of soil and possibly topography may have interacted to influence basicity. However, high Alsat values in some soil groups and horizons implies possibility of aluminium toxicity. This depends on the amount of Organic Matter (OM) in soil, since humic fractions reduces the phytotoxicity of Al3+ (Yamaguchi et al., 2004). In addition, Al-toxicity depends on the chemical form (Ginting et al., 1998). Increased exchangeable acidity suggests solubilization Al3+ which may not imply high activity of hydrogen ions in these soils (Styczen, 1992). Calcium-magnesium ratio was highest in soils derived form shale and least in those formed over lower coal measures (Table 5). The (Ca+Mg)/(Al+H) ratios were highest in alluvial soils and least in soils over upper coal measures. These elemental ratios are indicators to infer nutrient balance.
| Table 6: | Correlation coefficients between particle size distribution and exchangeable cations (p<0.05, n = 150) |
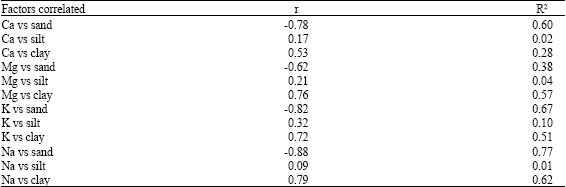 | |
A decrease of Ca/Mg ratio to a level below 3.0 results in unavailability of calcium and phosphorus (Landon, 1984). Low values of Ca + Mg) (Al + H) ratios indicates high values of acidic cations in the soils which portend poor nutrient availability in studied soils. Fairly high (Ca+Mg)/(Al+H) values in Alluvial soils (Table 5) could be due to marine depositions in the course of their pedogenesis. Significant relationships (p = 0.05) were observed between exchangeable basic cations and particle size fractions, especially sand and clay (Table 6). As sand content of soils increased, exchangeable basic cations decreased while the reverse was the case in the relationship between clay-sized fractions and basic cations Highly weathered soils of humid tropical environments have only modest amounts of plant-available Ca, Mg and K (Gillman et al., 1989) and this could be due to sand-clay contents and interactions in these soil. Good relationships between particle size fractions and exchangeable basic cations suggests their use for predicting nutrient availability as Ghosh and Singh (2001) recommended a consideration of clay mineralogy before application of K-fertilizers. However, the buffering characteristics of these basic cations in these soil groups should be investigated for increased understanding of their behaviour for sustained use in.
This study revealed differences in the distribution of basic cations among soil groups irrespective of similarity in climatic factors and land use history. Significant variations in basicity were also found among horizons and elemental ratios in the study site. There were good relationships between sand-and clay-sized fractions and basic cations, suggesting their usage in pedometric analysis of soils of the area for increased accuracy of models.