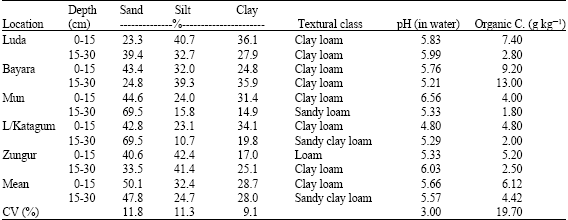
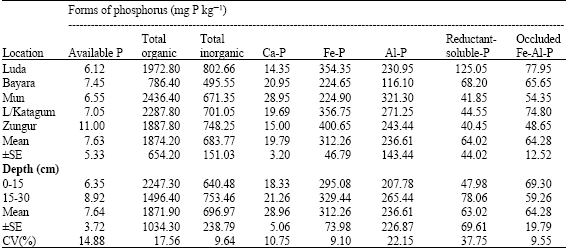
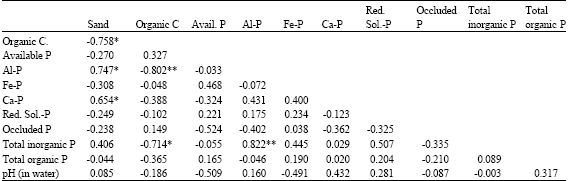
Research Article
Contents and Distribution of Phosphorus Forms in Some Haplic Plinthaquults in Bauchi Local Government Area, Bauchi State, Nigeria
Crop Production Programme, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi, Nigeria
S. I. Yerima
Crop Production Programme, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi, Nigeria
N. Voncir
Crop Production Programme, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi, Nigeria
B. I. Ahmed
Crop Production Programme, Abubakar Tafawa Balewa University, P.M.B. 0248, Bauchi, Nigeria