Research Article
Anticoccidial Effect of a Negilla sativa Seed-based Diet on Eimeria tenella Infection in Chickens
Department of Pathology and Poultry Diseases, Faculty of Veterinary Medicine, University of Kerbala, Iraq
LiveDNA: 964.16399
Coccidiosis is one of the most economically important diseases affecting commercial poultry1. Coccidiosis is related to intracellular protozoan parasites of the genus Eimeria and causes disruption of the intestinal epithelium, leading to diminished feed efficiency and loss of body weight1-3. Currently, in intensively reared poultry settings, this infection is controlled by the use of anticoccidial medications and/or by vaccination4. However, due to the increase in medication-resistant strains of Eimeria and rising public distress about medication remain in chicken products, additional treatments are urgently required5,6. Several studies have investigated the use of various dietary supplements and probiotics to control Eimeria infections7-10. Nigella sativa and its active constituents have been found to exhibit antioxidant activity, as well as immunomodulatory, immunotherapeutic potentials, antitoxic, anti-histaminic, anti-inflammatory properties and anti-helmenthic effects11,12.
This study evaluated the effects of Nigella sativa-based diets as natural anticoccidials through their antioxidant behavior to control E. tenella infections in broiler chickens.
After cleaning and disinfecting pens, 100 one-day-old broiler chicks (Ross 308) were randomly assigned to four equal groups of 25 chicks each. Group 1 (G1) was fed with a basal diet free of anticoccidial drug and supplemented with 1% whole crushed Nigella sativa seeds and Group 2 (G2) was fed with a basal diet containing salinomycin (Bio-Cox) at a rate of 60 g t–1 for 35 days. Group 3 (G3) and Group 4 (G4) were fed the basal diet only. G3 served as the challenged untreated control and G4 served as the untreated unchallenged control. G1, G2 and G3 groups were challenged with 1×104 mature oocysts of E. tenella. Nigella sativa crushed seeds were mixed daily with the basal diet for the duration of the experiment. Feed and water were provided ad libitum and continuous 24 h of lighting was applied. The environmental temperature was decreased gradually from 33°C on day 1 to 24°C (±1) on day 21. Chickens were vaccinated against Newcastle disease, infectious bronchitis and infectious bursal disease. To determine gut lesion scores, 6 chickens were randomly chosen from each group for scoring at day 7 after the Eimeria challenge13.
Eimeria infection and assessment of fecal oocyst production: The oocysts of a field isolate of E. tenella were obtained from a broiler farm that had chickens with bloody diarrhea, pathognomonic cecal lesions and a high mortality rate. Oocysts were collected from cecal contents and sporulated in a 2.5% potassium dichromate solution. Chickens in G1, G2 and G3 were orally challenged with 1×104 sporulated oocysts of E. tenella at 28 days of age. Fecal materials were collected from the litter at 10 days post-infection and the numbers of oocysts g–1 were assessed using a hemocytometer counting chamber14.
Blood sampling: Blood samples (2-3 mL) were taken from separate chickens (n = 6) at days 28 and 35 from the jugular vein and collected into tubes containing Ethylenediaminetetraacetic acid (EDTA) as an anticoagulant for the determination of glutathione (GSH) level, glutathione peroxidase (GSH-Px), superoxide dismutase (SOD) and catalase (CAT) activities in the hemolysate. Hemoglobin content (Hb) was performed as previously described by Jain15. Blood plasma was used for the estimation of malondialdehyde (MDA) as previously described by Ohkawa et al.16.
Hemolysate preparation: At 28 and 35 days of age, blood samples were collected in EDTA and centrifuged at 3000 rpm for 10 min; the buffy coat and plasma were separated from erythrocytes. Erythrocytes were washed three times with 0.9% normal saline and then 20% (v/v) hemolysate was prepared. The Beutler et al.17 procedure was used to measure hemolysate activities levels of GSH. GSH-Px was measured according to the method of Paglia and Valentine18, SOD was measured according to the method of Marklund and Marklund19 and CAT was measured according to the method of Aebi20.
Statistical analysis: The data were analyzed with SPSS 16.0 (SPSS Inc., Chicago, IL, USA) using one-way analysis of variance and least significance differences to determine the difference between groups at level of p<0.05.
Tables 1, 2, 3 and 4 show GSH levels, GSH-Px, SOD and CAT activities of the chickens at 28 and 35 days. From day 28, broilers from G1 group had the highest (p<0.05) GSH levels, GSH-Px, SOD and CAT activities. At day 35, G3 group had the lowest (p<0.05) GSH levels, as well as GSH-Px, SOD and CAT activities.
Significant levels of plasma MDA were apparent in G3 at day 35 only (Table 5).
| Table 1: | Levels of glutathione (GSH) in erythrocyte hemolysate in the different groups of chickens at 28 and 35 days of age |
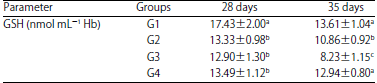 | |
| a,b,cp<0.05, SD: Standard deviation of means | |
| Table 2: | Levels of glutathione peroxidase (GSH-Px) activity in erythrocyte hemolysate in the different groups of chickens at 28 and 35 days of age |
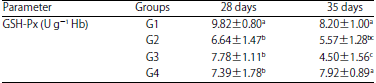 | |
| a,b,cp<0.05, SD: Standard deviation of means | |
| Table 3: | Levels of superoxide dismutase (SOD) activity in erythrocyte hemolysate in the different groups of chickens at 28 and 35 days of age |
 | |
| a,bp<0.05, SD: Standard deviation of means | |
| Table 4: | Levels of catalase (CAT) activity in erythrocyte hemolysate in the different groups of chickens at 28 and 35 days of age |
 | |
| a,b,cp<0.05, SD: Standard deviation of means | |
| Table 5: | Levels of malondialdehyde (MDA) in blood plasma in the different groups of chickens at 28 and 35 days of age |
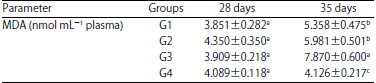 | |
| a,b,cp<0.05, SD: Standard deviation of means | |
Nevertheless, plasma MDA levels in the G1 and G2 groups were comparable. No significant differences were observed between all experimental groups at day 28.
The result of oocyst output at 28 and 35 days of age (10 days post-challenge infection) are presented in Fig. 1. The G3 group had the highest mean numbers of oocysts, with approximately 2.31×106 total oocysts shed. The G1 and G2 groups shed approximately 0.41×l06 and 0.34×106 total oocysts, respectively.
The lesion scores of broilers during the experimental period (mean±SD) are shown in Fig. 2. Lesion scores were zero at 28 days for all the groups before the challenge. After the challenge, the G3 group had the highest mean lesion scores with an average of 3.6, while the mean lesions scores for the G1 and G2 groups were 1.8 and 1.6, respectively.
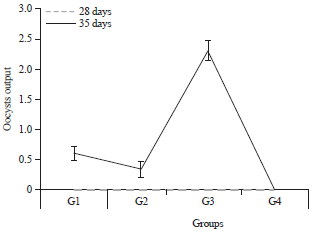 | |
| Fig. 1: | Oocysts output (×106) in the different groups of chickens at 28 and 35 days of age |
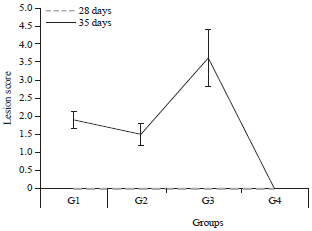 | |
| Fig. 2: | Lesion scores in the different groups of chickens at 28 and 35 days of age |
Reactive oxygen species (ROS) are molecules that are normally produced inside cells and tissues. The effects of ROS are usually mitigated by internal antioxidants and repair systems; however, the negative effects of ROS are not completely resolved and some of these effects accumulate over time to cause oxidative stress, a condition in which ROS production is greater than internal antioxidant systems21. Consequently, oxidative damage to different types of macromolecules can occur, which may be involved in the pathogenesis of many different diseases21,22.
Results from this study showed that supplementation with Nigella sativa seeds in the diet significantly increased levels of different antioxidants on day 28 (GSH, GSH-Px, SOD and CAT). The antioxidant properties of Nigella sativa seeds have been reported in many other previous studies23. It is found that up-regulation of these antioxidants in G1 was paralleled by a reduction in oxidative damage (MDA). These results are consistent with previous studies in which the oxidative damage (MDA) was significantly mitigated by enzymatic and non-enzymatic antioxidants24.
It is also found that the markers of antioxidants measured on day 35 were significantly higher in G1 compared to the other infected groups (G2 and G3), which led to decreased levels of oxidative damage (MDA) in both groups (G1 and G2). G3 had the lowest levels of antioxidants and highest levels of oxidative damage. These results were directly associated with the significant changes in the degree of lesions and the oocyte outputs among groups. G3 had the highest levels of lesions and oocyte outputs, while G2 and G1 had lower levels of lesions and oocyte outputs. The findings here highlight the effectiveness of herbal supplementation and the role of antioxidant properties of Nigella sativa seeds in recovery from parasitic infection. These seeds may be used in the diet instead of commercial chemicals like salinomycin.
Nigella sativa has an antioxidant capacity that can boost defense mechanisms of chickens against infection. This study recommends that Nigella sativa can be used commercially in the diet as a protective agent.
The author is grateful for the help of the laboratory staff and farm staff of the University of Baghdad who provided technical assistance during laboratory analysis and research.