ABSTRACT
Objective: The goal was to determine the frequencies and cytology variation (atypia) of basophils in caged hens at 18 and 77 weeks and the relation between basophils and heterophil/lymphocyte (H/L) ratios as stress measures. Methodology: Standard Differential Counts (SDC) were obtained from Wright stained blood films. When basophils were >5% of the total white blood cell count a two-tier Basophil Differential Count (BDC) was applied. As a first-stage, basophils were divided into “Resting” or “Reactive/atypical” types. When ~25% basophils were reactive or atypical, the second tier followed. One hundred metachromatic cells (basophils) were sorted as “Resting” or dendritic, dysgranulosis, dysplastic, dwarf, lake, mesomyelocytes, metamyelocytes, net, oncosis and toxics. Results: The study-wide basophil frequencies were ~3.5% of total leukocytes at either age. Basophil numbers were unaffected by cage styles, aviary (AV) conventional (CC) or enriched (EN). However, Total White Blood Cell Counts (TWBC) indicated leukocytosis (>25 K μL–1) and leukemoid reactions (>50 K μL–1) were common. The H/L ratios ~0.2 at 18 and ~0.3 at 77 weeks were below stress levels. Ages did not affect frequency of atypia but “Lake” and “Oncosis” basophils were more common at 77 weeks. “Basophilia” describes a sample SDC with >5% basophils, if >25% are non-resting/reactive or atypical types “basophiliosis” is applied. Conclusion: Atypical basophils are common in the blood of caged hens. Both basophilia and basophiliosis are likely inflammatory stress responses associated with Poly Microbial Bacteremia (PMB) and fungemia. A basophil differential count supplements the H/L ratio stress measure.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijps.2017.23.30
URL: https://scialert.net/abstract/?doi=ijps.2017.23.30
INTRODUCTION
Basophils are distinct granulocytes recognized microscopically by their metachromatic staining with Romanowsky dyes. Regarded as rare in the circulation by some authorities1-3, however, Maxwell et al.4 indicated otherwise. In the researchers experience, few avian blood samples are without basophils (Cotter, personal observation) and frequencies may range from 1% to as high as 23% of the TWBC5.
Use of a hemogram for assessing disease status is standard practice, applying blood data to determine stress expands its utility. Early observations on hens by Wolford and Ringer6 described how stressor agents affected differential leukocyte counts. Handling and cold exposure increased eosinophils but starvation had an opposite effect. Neither treatment affected basophils. However, injected ACTH, a stress hormone, increased heterophils and had a biphasic effect on basophils and eosinophils7. Basophilia resulted from the stress of prolonged food restriction4.
Injection of chicken wattles with human plasma resulted in localized swelling and increased circulating basophils8 an indication of Cutaneous Basophilic Hypersensitivity (CBH). Individual differences in the frequency of (circulating) basophils contributed to the variability of the swelling reaction to injected mitogen9. Alleles of the B-complex (chicken MHC) influenced the CBH response to Staphylococcus antigen10 suggesting basophil frequency is sensitive to bacteria. These observations were facilitated because metachromatic staining of basophils differentiates them from other granulocytes.
However, while resting basophils are metachromatic not all types fully display this property. Distinctive variation of cytoarchitecture among basophils is enough to warrant further differentiation. The study source material was blood obtained from the same commercial flock at placement (18 weeks) and at the end of production (77 weeks). Standard Differential Counts (SDC) identified samples with high basophil frequency and atypia. A Basophil Differential Count (BDC) followed by which "resting" and reactive/atypical types were distinguished. The latter were sorted as dendriform, dysgranulosis, dysplastic, dwarf, lake, mesomyelocytes, metamyelocytes, net, oncosis and toxic basophils. Examples of these cells are the subject. As basophils are inflammatory cells, both high frequency and atypia are likely consequences of bacteremia, fungemia and stress associated with life in cages. The present observations contribute to basic hematology, pathology and assessment of welfare.
MATERIALS AND METHODS
Hens: Lohmann white egg type hens (LSL) are the subjects. Those destined for conventional (CC) and enriched cages (EN) were raised in common until 18 weeks. At that time, they were placed in their final cage assignments at 6 per CC and 60 per EN. Those occupying aviaries (AV) at 850-1,700 hens per compartment had been there since day 1. Chicks were vaccinated against Marek’s disease and laryngotracheitis at the hatchery, additional spray vaccinations (NDV-bronchitis) were applied at 13 weeks, 5 weeks prior to collection of the first blood sample. Further, details regarding management, caging, etc., described by Cotter11.
Blood and stain procedure
Samples: About (1-3 mL) wing-vein blood was collected into tubes containing EDTA anti-coagulant. About ~3 μL was spread into monolayers on standard microscope slides. After drying in a hot air stream samples were post-fixed in 95% ethanol for 10-15 min. Staining was by Wright’s method following the manufacturer’s recommendations (Sigma Chemicals, St., Louis, Mo., procedure WSGD-128).
Statistics: Correlations (Pearson) t-tests (one and two-tail) and ANOVA were with Minitab® statistical software, release 17. Minitab, Inc., State College, PA. Significance is at 0.05.
Standard Differential Count (SDC): Candidate samples were identified by 2 preliminary Standard Differential Counts (SDC) of 200 leukocytes. If basophils exceeded 5% (basophilia) or were otherwise atypical, a basophil differential count followed BDC as described below. Leukocytes were sorted into categories: Small or medium lymphocytes (Ls, Lm) monocytes (Mn), heterophils (classic (HC), typical (HT) and variant (HV) types), basophils (Ba), blast cells (Bst) or eosinophils (Eo) by microscopic examination at 40x magnification. Morphological criteria are based on descriptions of Lucas and Jamroz12, Cotter11, Cotter and Bakst13 and Cotter and Heller14. Other sources consulted were Campbell and Ellis1 and Reagan et al.15.
Basophil Differential Count (BDC): All magenta (metachromatic) granulocytes of blood are considered as basophils. In a two-step process, basophils were classified as "resting" or "reactive/atypical" types. Reactive/atypical types were then sorted as dendriform, dysgranulosis, dysplastic, dwarf, lake, mesomyelocytes, metamyelocytes, net, oncosis and toxic cells. Representative examples of resting and selected atypical basophils are provided.
Light Microscopy and Photomicrographs: Olympus CX-41 (Olympus America, Center Valley, PA 18034-0610) equipped with Plan N 40x, 0.65 N.A. dry and Plan N, 1.25 N.A. 100x oil objectives. Photomicrographs: Images captured with an infinity-2 1.4 megapixel CCD USB 2.0 camera were processed with Infinity Analyze software (Release 6.5, Lumenera Inc., Ottawa, Ontario, CA K2E 8A7).
Heterophil lymphocyte ratios (H/L 1, H/L 2 and ΔH/L) and Total White Blood Counts (TWBC): Division of the sum of all heterophil types (HC, HT and HV) by the number of small "resting" lymphocytes (Ls) gives H/L 1. Division of the same heterophil value by the sum of all lymphocyte types, (Ls+Lm) gives H/L 2. The H/L 1 values will typically be greater than H/L 2. If all lymphocytes are Ls types H/L 2 = H/L 1. The H/L (1-2) difference (ΔH/L) should not exceed 0.1 in a non-stress environment14. Total White Blood Counts (TWBC) are estimates made from the SDC slides by a method described by Campbell and Ellis1.
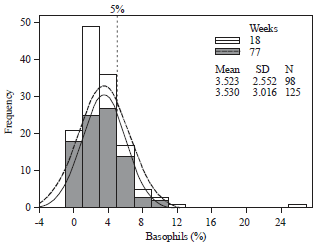 | |
| Fig. 1: | Normalized (%) circulating basophils for Lohmann (LSL) hens at 18 and 77 weeks, cages combined. The 5% basophilia threshold is indicated in solid line |
RESULTS
Population statistics: Table 1 and 2 contain population-wide SDC averages for 18 and 77 weeks hens. Heterophils (H) were not sub-typed at weeks 18 but at 77 weeks they were sub-typed as classic HC, typical HT or variant HV types. Cage styles did not affect H/L 1, H/L 2 or ΔH/L’s at weeks 18, nor do they indicate population-wide stress. However, medium sized lymphocytes (Lm) were twice as common (p>0.001) in AV samples as in CC or EN samples. As these "reactive" cells include plasmacytoid lymphocytes, plasmacytes and atypical lymphocytes16,17,13,14 a high frequency infers a cage effect not detected by standard analysis. This interpretation is supported by high TWBCs indicating both leukocytosis and leukemoid reactions were common at both ages (Table 1, 2).
Basophils occurred in 41/51 (80%) AV hens and 43/47 (91%) CC hens at 18 weeks among a population with low (non-stress) H/L ratios (Table 1). Basophils also occurred in the remaining samples, however they were found subsequent to completion of the SDC. A similar situation occurred at 77 weeks. Collectively, no sample was free of basophils.
At 77 weeks, neither the H/L values nor ΔH/L indicated population-wide stress (Table 2). However, as it was at placement, the ~12% Lm and high TWBC’s (~90 K, leukemoid reactions) indicate complex hemograms. The study-wide basophil averages of ~3.5% at both ages is less than the basophilia threshold (>5%) however many individual samples exceeded this level and in one case basophils composed ~26% of the TWBC (Fig. 1).
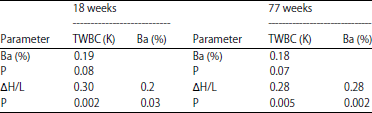
Population-wide correlations (cage styles combined) and probabilities indicate basophil (%) and TWBC (K) were weakly correlated at both ages (r2 ~0.18). Percent basophils and ΔH/L were somewhat correlated, r2 ~0.30. Raw basophil frequencies were not contributing to high TWBCs as much as other leukocytes.
| Table 1: | Study-wide SDCs (%) based on preliminary 200 cell counts, heterophil/lymphocyte statistics H/L 1, H/L 2, ΔH/L and TWBC (K) of hens at 18 weeks |
 | |
AV: Aviary, CC: Conventional, NS: Non-significant, H: Heterophil (unspecified type), L: Lymphocyte (small s, medium m), Bst: Granuloblast, Mn: Monocyte, Ba: Basophil, Eo: Eosinophil, H/L statistics: H/L 1 = H/Ls, H/L 2 = H/(Ls+Lm), ΔH/L = H/L (1-2) | |
| Table 2: | Study-wide SDC’s (%) based on preliminary 200 cell counts, heterophil/lymphocyte statistics H/L 1, H/L 2, ΔH/L and TWBC (K) of hens at 77 weeks |
 | |
AV: Aviary, CC: Conventional, EN: Enriched, NS: Non-significant, H: Heterophil, HC: Classic, HT: Typical, HV: Variant, L: Lymphocyte, S: Small, M: Medium, Bst: Granuloblast, Mn: Monocyte, Ba: Basophil, Eo: Eosinophil, H/L statistics: H/L 1 = (HC+HT+HV)/Ls, H/L 2 = (HC+HT+HV)/(Ls+Lm), ΔH/L = H/L (1-2) | |
Increases in the frequencies of all leukocytes (lymphocytes, granulocytes and monocytes) resulted in high TWBCs. The H/L 1 and H/L 2 ratios or their difference (ΔH/L) are independent of basophils or monocyte numbers and only the H/L 2 is affected by reactive/atypical lymphocytes. These observations emphasize the importance of integrating ratios with total cellularity to establish an accurate "blood picture". They further suggest basophilia and basophiliosis are useful as stress indicators independent of H/L’s. They are supported by an earlier report of doubling in basophil (%) by forced molting stress18 and the effect on basophil numbers by prolonged feed restriction stress19.
Basophil cytology: Dimensions of cells of Fig. 2-4 included in the legends are given as diameter D and perimeter P (μm) and area A (μm2). Basophil differential counts based on 100 cells (%) are in Table 3. Comparative data for resting, dwarf and oncosis basophil sizes in a single 77 weeks hen are in Table 4.
Resting: Resting basophils, the most common type (63%) are described first (Fig. 2a). They display a magenta (metachromatic) hue and contain a full complement of cytoplasmic granules. In some instances, granule density obscures the nucleus. The average diameter (D) of Wright stained resting basophils, ~9 μm is of similar size to toluidine-blue stained basophils described by Chand and Eyre5. An individual cell was assigned into the "resting" category as a default procedure if it could not otherwise be sorted.
Dendriform: A basophil having tree branch-like (dendriform) projections extending from the CM distinguishes this type (Fig. 2b). Those with clear dendriform projections are more common, in others, dendriform projections contain granules. At present, both types compose a single group.
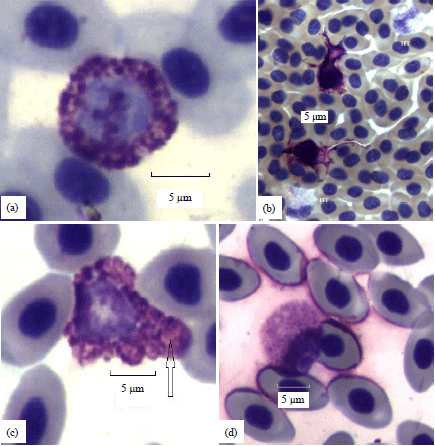 | |
| Fig. 2(a-d): | (a) A resting basophil has a central light blue nucleus and patchy chromatin in an 18 weeks hen, dimensions D 10, A 75, P 34 (~average study-wide). The fully stained magenta cytoplasmic granules ranging in size from ~0.1-1 μm are separated by clear spaces. Both nuclear and cytoplasmic membranes are entire, (b) The two dendriform basophils at the center (D 7.8 top, D 8 bottom) have branch-like projections extending 10 and 13 μm from CM. Two typical heterophils (HT) are at the top right and bottom left in an 18 weeks hen, (c) A distorted dysgranulocytic basophil of an 18 weeks hen (D 14, long axis) contains fewer, larger and irregularly sized cytoplasmic granules its nucleus is eccentric. The largest granule with a diameter of 2.4 μm (arrow) is twice the size of typical granules and (d) A dysplastic basophil of an 18 weeks hen (D 9.8, A 77, P 31) has an eccentric nucleus with an intact CM. The cytoplasm contains many small weakly stained granules |
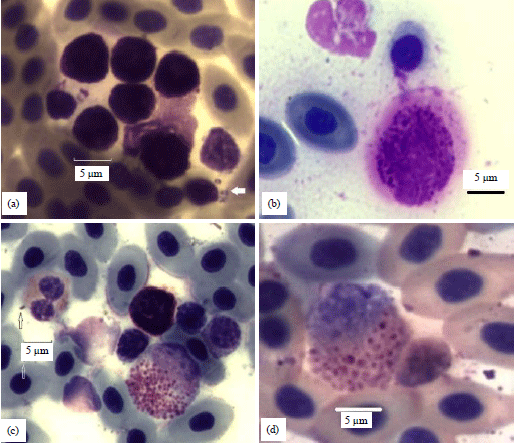 | |
| Fig. 3(a-d): | (a) A cluster of 6 dwarf basophils with an average D 5.9 (+/-0.4) in a 18 weeks hen. Several "hair-pin" bacilli are nearby (arrow), (b) A large lake-type basophil of a 77 weeks hen (D 12.1) with a translucent halo extending 1-3 μm beyond the CM contains a full complement of granules. Arrows locate several nearby bacteria, (c) A large (D 14.3, A 161 and P 45) developmental basophil of an 18 weeks hen (mesomyelocyte) has formed a rosette with a small lymphocyte. A resting basophil is nearby and a variant heterophil is to the left. A solitary bacillus is at the edge of the heterophil and an encapsulated coccus is located below (arrows) and (d) A large developmental basophil (metamyelocyte) (D 13, A 138 and P 41) of an 18 weeks hen with an eccentric nucleus has formed a rosette with a small lymphocyte at the right. The small cytoplasmic granules are faintly stained |
| Table 3: | Basophil differential counts (BDC%) and probability (P) for 18 and 77 weeks hens (cages combined) |
 | |
| Rst: Resting, Dnd: Dendriform, Dsg: Dysgranulosis, Dsp: Dysplastic, Dw: Dwarf, LK: Lake, Ms: Mesomyelocyte, Mt: Metamyelocyte, Nt: Net, Onc: Oncosis, Tx: Toxic | |
| Table 4: | Representative cell diameter sizes D (μm) and probability (P) from 77 weeks hen #520 |
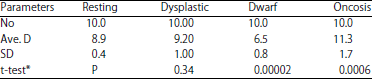 | |
*Average diameters of dwarf and oncosis cells were compared to resting cells with a "one-tail" t-test, a "two-tail" t-test was used to compare resting with dysplastic cells | |
Dysgranulosis types: Electron microscopy study of avian basophils has identified three granules: Dense, stippled and honeycomb types20. A similar repertoire occurs in the related mast cell where granules of all types have an average diameter of 0.7 μm (Cotter, personal observation). The granule diameters of resting basophils (Fig. 2a) average 0.8 (+/-0.1) μm as measured by light microscopy (Cotter, personal observation). Larger sized and irregular granules occur in dysgranulosis, those of Fig. 2 caverage 1.7 (+/-0.4) μm, some granules have faintly stained cores with fully stained edges.
Dysplastic types: Basophil granules contain histamine and other pharmacologically active substances as reviewed in Van Beek et al.21.
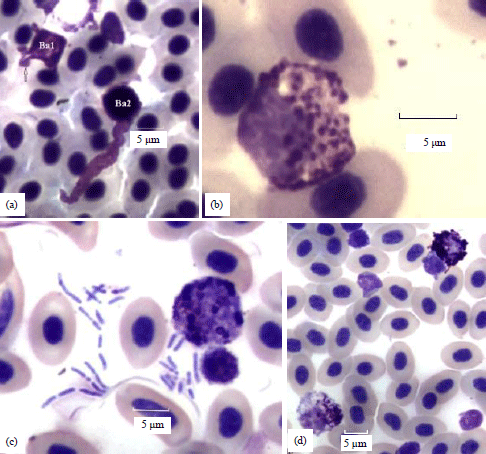 | |
| Fig. 4(a-d): | (a) Two dwarf net-type basophils (Ave. D 6.4) in an 18 weeks hen. Net processes are at minimal lengths in Ba1 at the top left (arrow). The cell at the bottom right (Ba2) has a solitary net process of 18 μm, (b) A slightly swollen cell (oncosis, D 10.6) in an 18 weeks hen, the nuclear and cytoplasmic membranes are intact, the nucleus is eccentric. The cytoplasmic granules are separated from each other by an increase in perigranular spacing, (c) A toxic basophil metamyelocyte has formed a rosette with a small lymphocyte. The field contains 25 bacilli ranging in length from 1.5-5.2 μm and (d) A large toxic basophil (D 11) with atypical bluish cytoplasm (bottom left) and an eccentric nucleus. Its sparse granules are restricted to one side of the cell. A reactive basophil with small projections of the CM (filopodia, arrows, top right) has formed a rosette with a small lymphocyte |
| Table 5: | Correlations/probability (P) among percentage of basophils (Ba) as determined by SDC, ΔH/L (H/L 1-H/L 2) and total white cell count, TWBC (K) at 18 and 77 weeks |
 | |
Abnormalities of reduced granule size or quality of active substance (dysplasia) can result in alteration of metachromatic staining. Granule resolution allowed by light microscopy, however, may not differentiate all dysplastic types. When the abnormality involves multiple granules (full dysplasia), the cytoplasmic stain is faint (Fig. 2d). Cell numbers 191 and 192 illustrated in Lucas and Jamroz12 resemble dysplastic types.
Dwarf: Dwarf types ranged from 0-37% of the study-wide BDCs (Fig. 3a). The average diameter of 12 dwarf basophils (7.3 μm) was significantly smaller (p = 0.0002) than 12 normal sized cells (9.4 μm) in the same 18 weeks hen. Dwarf basophils may display other forms of atypia however such as dendriform projections or laking but they were sorted as dwarf types alone. Table 5 contains size comparisons using data from a 77 weeks hen.
Lake types: Diffusion of cytoplasmic substance from an otherwise intact cell resulting in a halo describes the lake-type atypia. As in oncosis, the CM should remain intact and the cell should retain a full complement of granules (Fig. 3b). Lake types were more common at 77 weeks (Table 3).
Mesomyelocyte and metamyelocyte developmental types: Basophil development occurs in bone marrow, a circulating developmental cell (mesomyelocyte or metamyelocyte) should not be in blood in the absence of stress or disease. Thus, the circulating cells of Fig. 3 are highly atypical. A large (D 14.6 μm, A 167 μm2, P 46 μm) mesomyelocyte with small (Ave. <0.6 μm) widely separated cytoplasmic granules was in an 18 weeks hen. The cell has formed a rosette with a small lymphocyte (Ls) located at the 1 o’clock position (Fig. 3c). Mature resting basophils rarely form rosettes with other cells. A smaller (D 10 μm) more developmentally advanced metamyelocyte/Ls rosette has larger and more consolidated granules was found in another 18 weeks hen (Fig. 3d).
Net types: Basophils having thin diffuse streams of granule-free cytoplasmic substance extending beyond the CM characterize the net type. They differ from dendriform types whose extensions are thicker and in the form of branches (Fig. 4a).
Oncosis: Swollen (oncosis) basophils recognized by larger size and greater perigranular spacing were more common at 77 weeks (Table 3). In true oncosis (Fig. 4b) both nuclear and CM remain intact otherwise "smudge-cell" artifacts may be mistaken for oncosis. Due to swelling, the diameter of a typical oncosis cell was ~2 μm greater than resting types (Table 5). Cell No. 194 of Lucas and Jamroz12 resembles the oncosis types described here.
Toxic changes: Basophils with unusual hues, blue vs., magenta staining, cytoplasmic vacuoles, or unequal distributions of granules represent this study. They often formed rosettes with small lymphocytes, usually indicating the cell is a developmental stage, either a mesomyeocyte or a metamyelocyte (Fig. 4c, d).
DISCUSSION
The present observations indicate basophils are common in hen blood, occurring at ~3.5% of total leukocytes. This frequency occurs in the context of high total white cell counts often at leukocytosis (>25 K μL–1) or leukemoid reaction (>50 K μL–1) levels, a characteristic of complex hemograms. Distinctive cytology differentiates "resting" from "reactive/atypical" and developmental types. The cytoplasm of resting basophils is filled with small (D ~0.8 μm) metachromatic granules at times so densely packaged as to obscure the nucleus. The differences among various forms of reactive/atypical cells are enough to allow differentiation by light microscopy. The categories are developmental, dwarf, dysgranulosis, dysplastic, lake, net, oncosis and toxic types. These distinctions, useful as sorting conveniences, might impose artificial discontinuity and cells can display multiple atypia.
Dendriform basophils have characteristic tree branch-like elaborations of the CM at times containing granules. Large or irregularly shaped and aggregated granules either deeply or faintly stained characterize the dysgranulosis types. Dwarf basophils (D<7 μm) are a heterogeneous group sometimes exhibiting atypia in additional to a small size. Lake types have clear halos, a full complement of granules and an intact CM.
Early (mesomyelocyte) and later developmental forms (metamyelocytes) often forming rosettes with other lymphocytes are highly unusual in the circulation. Finding one or two during a SDC is remarkable by itself. The cytoplasmic elaborations of net-type basophils are generally free of granules and distinct from dendriform basophils. Oncosis and lake types are larger than resting cells but because of different reasons. Cell swelling occurs in oncosis but granules remain within the intact CM.
Atypical basophils do not occur in the absence of other unusual hematology. They are consistent in blood samples also positive for bacteria and fungi. In vitro study of human basophils indicates differential activation of the granules by the tumor promoter 12-0-tetradecanoyl-phorbol1 3-acetate (TPA) and the bacterial peptide Formyl Methionyl Leucyl Phenylalanine (FMLP). Activation of granules by histamine begins in seconds and continues over many minutes in basophils exposed to TPA. The FMLP activation follows different kinetics described by Dvorak et al.22. Perhaps some basophil atypia described here corresponds to what occurs in human cells. Exposure of basophils to substances or metabolites of bacterial and fungal origin might account for both basophilia and basophiliosis. They may parallel the experimental results affecting basophils using staphylococcal and human plasma antigens10,8.
CONCLUSION
In conclusion, basophilia and basophiliosis are indicative of complex hemograms and provide an additional stress measure independent of H/L ratios and the TWBC. To the author’s knowledge, this study is the first attempt to differentiate basophils by light microscopy.
SIGNIFICANCE STATEMENT
Basophils and atypical basophils are common in the blood of caged hens often occurring at frequencies ranging from 3-5% of total white blood cells. Both basophilia (a high absolute basophil count) and basophiliosis (a high frequency of atypical cells) are indicative of complex hemograms. They are likely a consequence of inflammatory stress responses associated with poly microbial bacteremia and fungemia. They provide a stress measure independent of heterophil/lymphocyte ratios. This study is the first demonstration of differentiation of basophils by light microscopy. The present observations contribute to basic hematology, pathology and assessment of welfare.
REFERENCES
- Campbell, T.W., C.K. Ellis and B. Doneley, 2007. Avian and Exotic Animal Hematology and Cytology. 3rd Edn., Blackwell Publishing, Ames, Iowa, USA, Pages: 270.
Direct Link - Maxwell, M.H., G.W. Robertson, S. Spence and C.C. McCorquodale, 1990. Comparison of haematological values in restricted-and ad libitum-fed domestic fowls: White blood cells and thrombocytes. Br. Poult. Sci., 31: 399-405.
CrossRefPubMedDirect Link - Chand, N. and P. Eyre, 1978. Rapid method for basophil count in domestic fowl. Avian Dis., 22: 639-645.
CrossRefDirect Link - Wolford, J.H. and R.K. Ringer, 1962. Adrenal weight, adrenal ascorbic acid, adrenal cholesterol and differential leucocyte counts as physiological indicators of stressor agents in laying hens. Poult. Sci., 41: 1521-1529.
CrossRefDirect Link - Newcomer, W.S., 1957. Blood cell changes following ACTH injection in the chick. Proc. Soc. Exp. Biol. Med., 96: 613-616.
PubMed - Cotter, P.F. and T. Wing, 1987. Kinetics of the wattle reaction to human plasma. Avian Dis., 31: 643-648.
CrossRefDirect Link - McCorkle, Jr. F., I. Olah and B. Glick, 1980. The morphology of the phytohemagglutinin-induced cell response in the chicken's wattle. Poult. Sci., 59: 616-623.
CrossRefDirect Link - Cotter, P.F., R.L. Taylor Jr., T.L. Wing and W.E. Briles, 1987. Major histocompatibility (B) complex-associated differences in the delayed wattle reaction to staphylococcal antigen. Poult. Sci., 66: 203-208.
CrossRefDirect Link - Cotter, P.F., 2015. An examination of the utility of heterophil-lymphocyte ratios in assessing stress of caged hens. Poult. Sci., 94: 512-517.
CrossRefDirect Link - Cotter, P.F. and M.R. Bakst, 2016. A comparison of Mott cell morphology of three avian species. II.-Bad behavior by plasmacytes? Poult. Sci., (In Press).
CrossRefDirect Link - Cotter, P.F. and E.D. Heller, 2016. Complex hemograms of isolator raised Specific Pathogen Free (SPF) chicks. Int. J. Poult. Sci., 15: 211-217.
CrossRefDirect Link - Cotter, P.F., 2015. Are peripheral Mott cells an indication of stress or inefficient immunity? Poult. Sci., 94: 1433-1438.
CrossRefDirect Link - Cotter, P.F., 2015. Atypical lymphocytes and leukocytes in the peripheral circulation of caged hens. Poult. Sci., 94: 1439-1445.
CrossRefPubMedDirect Link - Brake, J., M. Baker, G.W. Morgan and P. Thaxton, 1982. Physiological changes in caged layers during a forced molt. 4. Leucocytes and packed cell volume. Poult. Sci., 61: 790-795.
CrossRefDirect Link - Maxwell, M.H. and G.W. Robertson, 1995. The avian basophilic leukocyte: A review. World's Poult. Sci. J., 51: 307-325.
CrossRefDirect Link - Maxwell, M.H., 1973. Comparison of heterophil and basophil ultrastructure in six species of domestic birds. J. Anatomy, 115: 187-202.
PubMedDirect Link - Van Beek, A.A., E.F. Knol, P. De Vos, M.J. Smelt, H.F.J. Savelkoul and R.J.J. Van Neerven, 2012. Recent developments in basophil research: Do basophils initiate and perpetuate type 2 T-helper cell responses? Int. Arch. Allergy Immunol., 160: 7-17.
CrossRefDirect Link - Dvorak, A.M., D.J. MacGlashan Jr., E.S. Morgan and L.M. Lichtenstein, 1996. Vesicular transport of histamine in stimulated human basophils. Blood, 88: 4090-4101.
Direct Link








