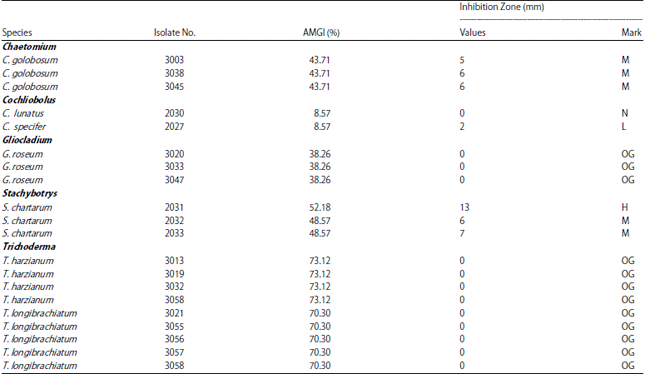
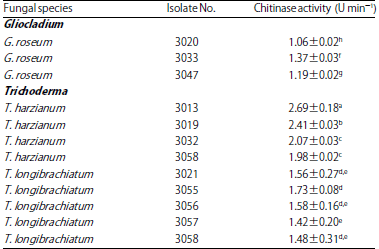
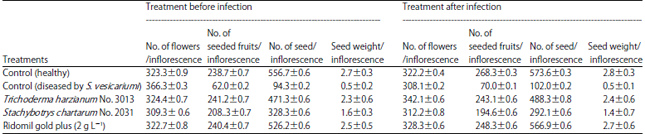
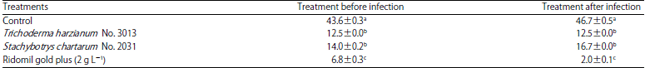Research Article
Control of Stemphylium Leaf Blight Disease of Onion and Elevation of Seed Production Using Certain Bioagents
Department of Plant Pathology, Faculty of Agriculture, Assiut University, 71526 Assiut, Egypt
LiveDNA: 20.1102
Sobhy I.I. Abdel-Hafez
Department of Botany and Microbiology, Faculty of Science, Assiut University, 71516 Assiut, Egypt
Ismail R. Abdel-Rahim
Department of Botany and Microbiology, Faculty of Science, Assiut University, 71516 Assiut, Egypt
LiveDNA: 20.13572