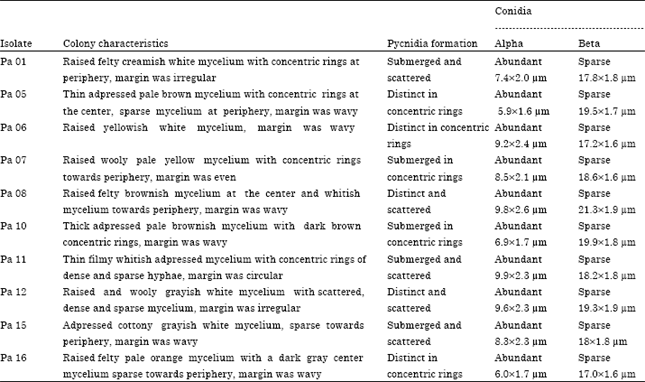
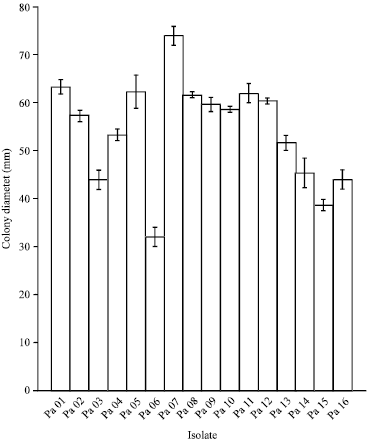
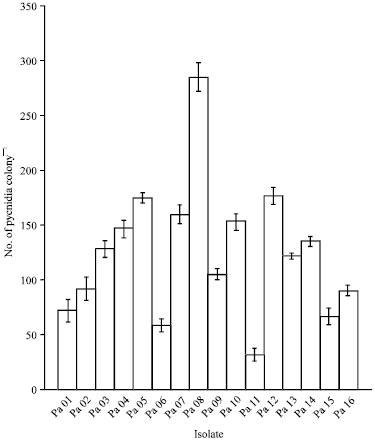
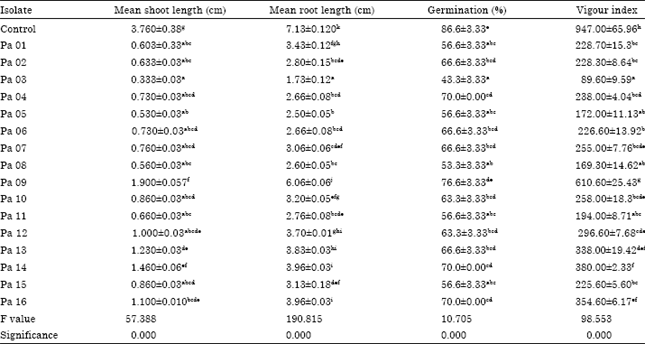
Research Article
Cultural, Morphological and Biochemical Variability among the Isolates of Phomopsis azadirachtae from Karnataka
Post Graduate Department of Microbiology, Maharani�s Science College for Women, JLB Road, Mysore-570005, Karnataka, India
S. Shankara Bhat
Department of Studies in Microbiology, Manasagangotri, University of Mysore, Mysore-570006, Karnataka, India
K. Girish
Post Graduate Department of Microbiology, Maharani�s Science College for Women, JLB Road, Mysore-570005, Karnataka, India