ABSTRACT
Background and Objective: Obesity is one of the main comorbidities impeding cardioprotection in patients with Ischemia Reperfusion (IR) injuries. This study investigated the combined postconditioning effect of melatonin and sitagliptin on myocardial IR-induced arrhythmias in obese rats by evaluating Nitric Oxide metabolites (NOx), oxidative stress and the involvement of the mitochondrial ATP-sensitive potassium channels (mitoK-ATP). Materials and Methods: Sixty-six Sprague-Dawley rats were fed a high-fat diet for 8 weeks to induce obesity. For IR injury modelling, the left coronary artery was ligated for 30 min to induce myocardial ischemia, followed by 2 hrs reperfusion. Melatonin (10 mg kg–1) and sitagliptin (20 mg kg–1) were intraperitoneally administered to obese rats early in reperfusion. Electrocardiography was recorded thoroughly and the Lambeth convention was employed to interpret myocardial arrhythmias (ventricular premature complexes, tachycardia and fibrillation). NOx, oxidative stress markers and mitochondrial function were evaluated using the Griess method, spectrophotometry and fluorometry. Results: Individual application of melatonin or sitagliptin reduced overall arrhythmogenic effects of IR injury in obese rats, however, the effect of their combination was greater on reducing the number, duration, incidence and severity of arrhythmias. The combination of melatonin and sitagliptin also increased the production of NOx, glutathione-peroxidase, superoxide-dismutase and catalase, while reducing malondialdehyde following IR injury. Mitochondrial depolarization and ROS production were also significantly reduced by postconditioning modalities as compared to non-treated IR-experienced obese rats. Interestingly, blockade of mitoK-ATP channels via 5-hydroxydecanoate significantly abolished melatonin/ sitagliptin-induced cardioprotection. Conclusion: Combination of melatonin/sitagliptin conferred considerable anti-arrhythmic influences against myocardial IR injury in obese rats through modulation of mitoK-ATP/NO/ROS signalling.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijp.2022.657.666
URL: https://scialert.net/abstract/?doi=ijp.2022.657.666
INTRODUCTION
Obesity is a global epidemic that imposes very serious consequences on the cardiovascular system, predisposing to ischemic heart disease1. Growing evidence suggests that obese patients have a greater susceptibility to myocardial Ischemia Reperfusion (IR) injury and most importantly, cardioprotection by routine modalities is usually lost in the presence of obesity conditions2. The underlying mechanisms responsible for this resistance to cardioprotection have not been explained and warrant further elucidation. IR-induced myocardial arrhythmias are common clinical manifestations in patients with myocardial infarction, whose severity and incidence increase in the presence of hypercholesterolemia and obesity. The arrhythmogenic complications of obesity in myocardial IR are diverse and may include impairment in the balance of oxidative stress and antioxidant capacities, increased inflammation, coronary calcification, cardiac dysrhythmia and electrical dysfunction, endothelial dysfunction, over-activation of renin-angiotensin and sympathetic nervous systems1.
Adipose tissue in obese subjects is determined as an important source of Reactive Oxygen Species (ROS)3. Overproduction of ROS may contribute to the development of elevated lipid peroxidation and oxidative stress, leading to an imbalance in electrolyte and ionic homeostasis and dysregulation of vital organelles like mitochondria. Mitochondrial ATP-sensitive potassium (mitoK-ATP) channels are suggested to be a critical effector for the protection of IR myocardium and the inhibition of IR-induced ventricular arrhythmias4. These channels mediate the protective effects of preconditioning and postconditioning and an important endogenous mediator, Nitric Oxide (NO). Although physiologic ROS signal is required for cardioprotection induced by preconditioning, closure of mitoK-ATP channels during reperfusion facilitates mitochondrial dysfunction and thereby ROS overproduction, leading to exacerbation of IR injury5. Thus, targeting these channels by powerful and safe interventions can alleviate the obesity-induced oxidative attacks during IR injury.
It has been proposed that the efficacy of melatonin increases when combined with Dipeptidyl Peptidase-4 (DPP-4) inhibitors in metabolic and diabetic conditions6,7 but it is yet unknown whether their individual or combination use can induce full cardioprotection in the setting of obesity. As a multifaceted endogenous hormone, melatonin regulates circadian rhythms and has strong antioxidative and anti inflammatory potentials sufficient to protect the myocardium against IR insults8,9. Interestingly, melatonin hindered the mitochondrial permeability transitions-mediated mitochondrial dysfunction and cellular oxidative stress through the opening of mitoK-ATP channels in the brain10. Sitagliptin is an important antidiabetic drug that belongs to the DPP-4 inhibitors class and has obtained great attention to repurposing it in cardiovascular medicine because of having beneficial cardioprotective effects11,12. It also decreased leptin and visfatin levels increased adiponectin levels and reduced the occurrence of hypoglycemia in obese patients13. Likewise, the positive impacts of sitagliptin on reduction of cardiomyopathy biomarkers and improvement of the electrocardiographic and hemodynamic function of the diabetic heart were significantly increased when combined with trigonelline14. Still, sitagliptin did not reduce the risk of cardiovascular death or hospitalization for heart failure in patients with diabetes15. These findings rationalize the concomitant use of sitagliptin with other drugs in reducing the myocardial IR complications of cardiovascular comorbidities.
Therefore, according to the beneficial potentials of both melatonin and sitagliptin, this study aimed to assess the hypothesis that postconditioning with melatonin and sitagliptin can confer substantial antiarrhythmic effects following myocardial IR injury in obese rats by modulating NO production, mitochondrial function and oxidative stress and whether the mitoK-ATP channels are involved in the cardioprotective effect of melatonin/sitagliptin combination.
MATERIALS AND METHODS
Study area: The study was carried out at the Department of Emergency, Capital Medical University, China from January-October, 2021.
Animals and materials: In this study, 36 male Sprague-Dawley rats (250±20 g) were enrolled and kept in the animal room under standard conditions of 12 hrs light/dark cycle at 22±2°C and 50-55% humidity. The rats were fed a high-fat diet containing 45% fat, 20% protein and 35% carbohydrate, for 8 weeks. All experimentations followed international animal research ethical standards, approved by the University Ethics Committee under the approval code of 2021-BJ-3208). Melatonin, Griess reagents, JC-1 and DCFDA dyes and 5-hydroxydecanoic acid were bought from Sigma-Aldrich (St. Louis, MO, USA). Sitagliptin was purchased from Novartis, (Basel, Switzerland). The assay kits for Creatine Kinase (CK), Malondialdehyde (MDA), catalase, Superoxide Dismutase (SOD) and Glutathione Peroxidase (GPX) were obtained from Jiancheng Bioengineering Institute (Nanjing, China).
Myocardial in vivo IR injury: Eight weeks after feeding a high-fat diet, rats were anaesthetized with sodium pentobarbital (50 mg kg–1), intraperitoneally and then subjected to thoracotomy by an incision through the 4th and 5th intercostal muscles. After exposing the hearts, the Left Anterior Descending (LAD) coronary arteries were ligated with a 5-0 silk thread to induce left ventricular ischemia. After 30 min of ischemia, the LAD was reopened and reperfusion was established for 120 min.
Experimental design: The obese rats were randomly divided into 6 groups with 6 rats in each:
- Control group, received only thoracotomy without LAD ligation
- IR group, received IR injury via LAD ligation and reopening
- IR+Mela group, received IR injury and melatonin (10 mg kg–1, i.p.)16, 5 min before the onset of reperfusion
- IR+Sita group, received IR injury and sitagliptin (20 mg kg–1, i.p.)11, 5 min before the onset of reperfusion
- IR+Mela+Sita group, received IR injury and both drugs
- IR+5HD+Mela+Sita group, received IR injury and both drugs as well as 5-hydroxydecanoic acid, (5HD, 100 μm, i.p.)17, 10 min before the onset of reperfusion
The rats in control and IR groups were injected with the same volumes of distilled water, i.p. The dosages of melatonin, sitagliptin and 5HD used in this study were selected according to the most protective doses reported in previous studies.
Recording and interpretation of myocardial arrhythmias: During the IR experiments, 3 golden electrodes were attached to both right and left hands and the right foot of the rats to record electrocardiographic traces on the axis lead 2. During the reperfusion phase, the count and duration of ventricular arrhythmias including Premature Ventricular Complexes (PVC), Ventricular Tachycardia (VT) and Ventricular Fibrillation (VF), the incidence of VT and VF and the severity (scoring) of arrhythmias were evaluated based on the Lambeth convention (Fig. 1).
Measurement of nitric oxide metabolites: Myocardial contents of Nitric Oxide metabolites (NOx) were measured by the Griess reaction.
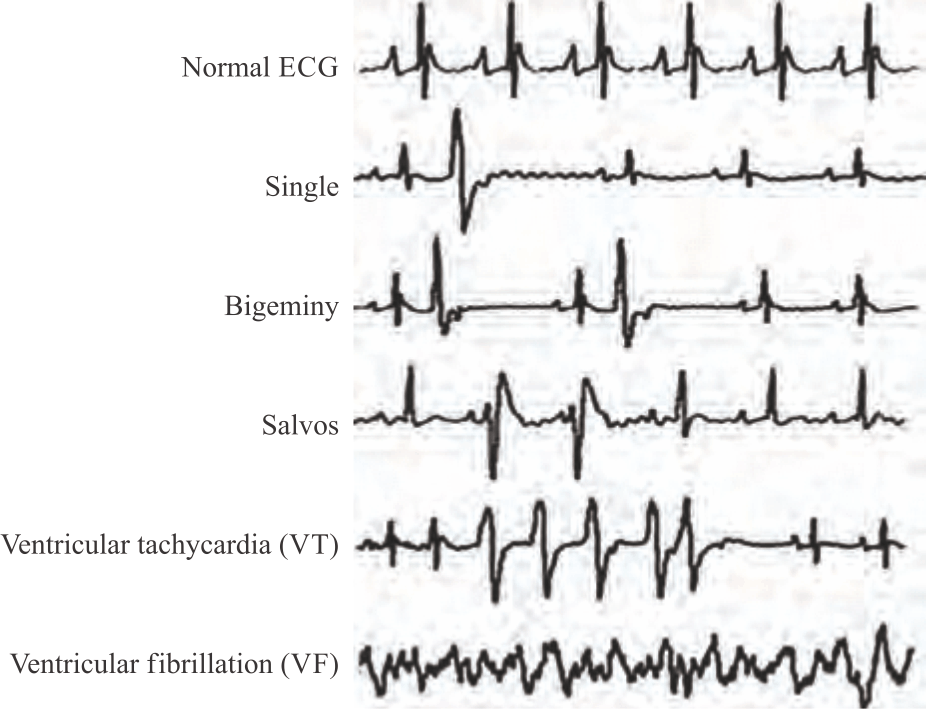 |
| Fig. 1: | Tracing of myocardial arrhythmias types according to the lambeth convention PVC: Premature ventricular complexes, VT: Ventricular tachycardia and VF: Ventricular fibrillation, single beats, bigeminy and salvos are considered PVCs |
Briefly, left ventricular samples were deproteinized by zinc sulfate (15 mg mL–1) and then, a 100 μL of the resultant supernatant was transferred to a microplate well, followed by the addition of 100 μL vanadium (3) chloride to each well to reduce nitrate to nitrite. Thereafter, 50 μL from each of Griess reagents including 2% sulfanilamide and 0.1% N-ethylenediamine dihydrochloride were added and the wells were incubated at 37°C. After 30 min, the absorbance of samples was read at 540 nm using the ELISA reader and the NOx levels were expressed as nmol mg–1 of protein.
Quantification of oxidative stress markers and CK: At the end of reperfusion time, the blood samples were obtained from the heart of rats and then the hearts were removed from the body and the left ventricles were isolated immediately. Then, the samples from the ischemic zone of the left ventricles were frozen by liquid nitrogen and subjected to homogenization and subsequent centrifugation at 13,000×g for 10 min at 4°C. The supernatants were taken and used for measurement of oxidative stress marker MDA by thiobarbituric acid reaction and endogenous antioxidative enzymes catalase, SOD, GPX, using the spectrophotometric technique, based on the instruction of the kits provided by the manufacturer. The protein content of samples was determined through the Bradford method18 and the values of antioxidant and oxidative stress markers were normalized with the protein content of each sample. Blood samples were used for the measurement of CK levels using the ELISA kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The absorbance of the samples was measured at 340 nm and the values were reported as U L–1.
Extraction of mitochondrial fraction: The left ventricular samples from ischemic areas were fresh homogenized in isolation buffer A, containing 0.3 mmol Phenyl-Methyl-Sulfonyl Fluorides (PMSF), 1 Na3VO4, 1 mmol NaF, 1 mmol EDTA, 10 mmol Tris-HCl, 250 mmol sucrose and protease inhibitor. After serial centrifugation of solution as 1000×g and 10000×g, the pellets were suspended again in buffer B, containing 0.3 mmol PMSF, 1 mmol NaF, 1 mmol Na3VO4, 10 mmol EDTA, 20 mmol Tris-HCl, 150 mmol NaCl, 1% NP-40 and protease inhibitor and centrifuged at 21000×g for 10 min. The supernatants, the mitochondrial fractions, were obtained and used for mitochondrial studies.
Determination of mitochondrial ROS: For the determination of mitochondrial ROS, mitochondrial supernatants were incubated with 2 μM Dichlorodihydro-Fluorescein Diacetate (DCFDA) dye for 30 min at 37°C. The solution was excited at 480 nm and the emission was recorded at 530 nm, by fluorometry and a fluorescent reader (Molecular Devices, San Jose, CA, USA). The fluorescence intensity of each sample was calibrated according to the protein amount of the supernatant and the value was expressed as mitochondrial ROS level.
Estimation of mitochondrial membrane potential: JC-1 staining method19 was employed to measure the changes in mitochondrial membrane potential (ΔΨ) of the myocardial samples according to the manufacturer’s instructions. Briefly, the mitochondrial supernatants were suspended in 2 mL of warmed phosphate-buffered saline containing 2 μL of JC-1 and incubated for 30 min at 37°C in a dark place. After 3 washes with phosphate-buffered saline, the JC-1 fluorescence of the samples was measured using a fluorometer (Molecular Devices, San Jose, CA, USA).
Statistical analysis: The data was reported as Mean±SD. The changes between different groups were analyzed by one-way ANOVA, followed by the Tukey test as a post hoc. A p<0.05 was considered a significant difference.
RESULTS
Melatonin/sitagliptin combination reduced IR-induced ventricular arrhythmias in obese rats, which reversed by 5HD: Induction of IR injury in obese rats significantly increased the count and timing of PVC (p<0.001), VT (p<0.01) and VF (p<0.05 and p<0.01) arrhythmias as compared with obese rats in the control group experiencing no IR injury (Fig. 2a-b). Melatonin alone significantly reduced the count and timing of arrhythmias, whereas sitagliptin did not affect the count of PVC and VT and the duration of VT. However, the combination of melatonin and sitagliptin significantly and more effectively reduced all arrhythmias as compared with the IR group. Simultaneous inhibition of mitochondrial K-ATP channels by 5HD significantly abolished the effects of combination therapy on the count and timing of PVC, VT and VF (p<0.05-p<0.001) (Fig. 2a-b).
Following IR induction in obese rats, the incidence of VT and VF (Fig. 3a) as well as the severity (scoring) of arrhythmias (Fig. 3b), was considerably increased as compared with the control group (p<0.001). Individual or combined administration of melatonin and sitagliptin at the reperfusion phase significantly reduced these parameters in comparison to the IR group but the effect of combination therapy was greater than those of individual treatments. Administration of 5HD, although did not have a significant effect on cardiac arrhythmias alone, significantly inhibited the protective effect of the combination of melatonin plus sitagliptin on the incidence of these arrhythmias and scoring, compared with the IR+Mel+Sit group (p<0.01 and p<0.001) (Fig. 3a-b).
Melatonin/sitagliptin combination reduced IR-induced CK release in obese rats, which reversed by 5HD: Myocardial CK was significantly increased following IR injury in obese rats and administration of melatonin and sitagliptin considerably reduced IR-induced increase in CK release (Fig. 4). Importantly, the reduction of CK by combination therapy was significantly greater than the individual treatments (p<0.05). However, 5HD co-treatment significantly suppressed the combination effect on CK level (p<0.01).
Melatonin/sitagliptin combination reduced IR-induced myocardial oxidative stress in obese rats, which reversed by 5HD: Intracellular concentrations of MDA as the main marker of lipid peroxidation and catalase, SOD and GPX the main endogenous antioxidant enzymes were measured to quantify the oxidative stress (Fig. 5a-d). IR injury induction in obese rats significantly increased the amount of MDA and significantly lowered the amounts of catalase, SOD and GPX as compared to the control group (p<0.001). Melatonin alone did not affect catalase but reduced MDA (p<0.05) and increased SOD and GPX (p<0.05) in comparison to the IR group. In addition, sitagliptin alone did not affect MDA, catalase and GPX levels but only increased SOD level (p<0.05).
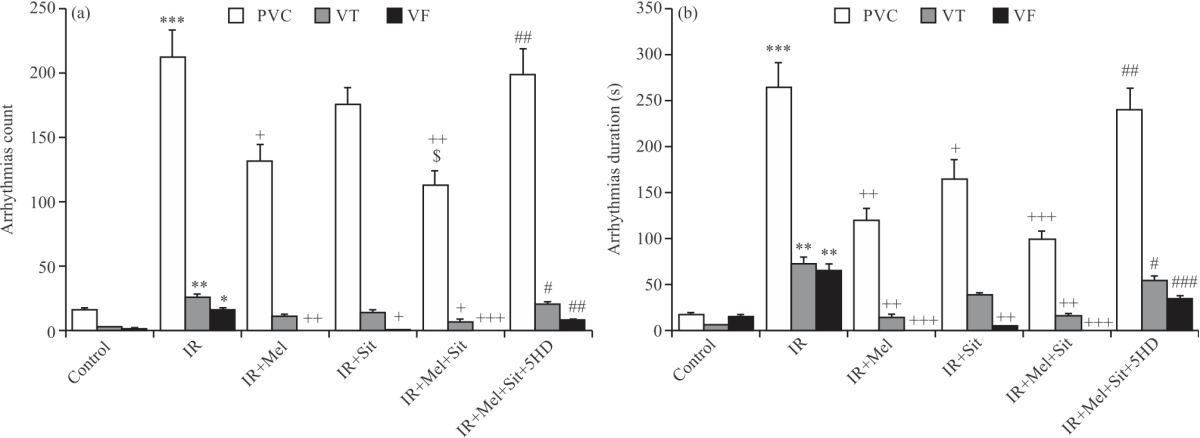 |
| Fig. 2(a-b): | Number and duration of myocardial arrhythmias in experimental groups, (a) Number and (b) Duration of myocardial IR-induced arrhythmias Mean±SD and n = 6. IR: Ischemia reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, PVC: Premature ventricular complexes, VT: Ventricular tachycardia and VF: Ventricular fibrillation, *p<0.05, **p<0.01, ***p<0.001 vs. Control group, +p<0.05, ++p<0.01, +++p<0.001 vs. IR group, #p<0.05, ##p<0.01, ###p<0.001 vs. IR+Mel+Sit group and $p<0.05 vs. IR+Sit group |
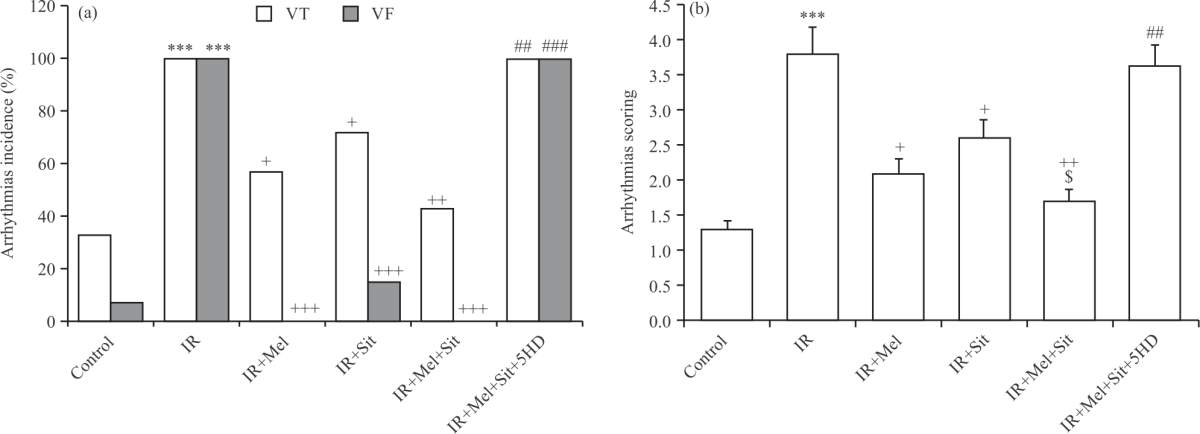 |
| Fig. 3(a-b): | (a) Incidence of VT, VF and (b) Severity of myocardial arrhythmia, IR-induced arrhythmia in experimental groups Mean±SD and n = 6. IR: Ischemia-reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, VT: Ventricular tachycardia and VF: Ventricular fibrillation, ***p<0.001 vs. control group, +p<0.05, ++p<0.01, +++p<0.001 vs. IR group, ##p<0.01, ###p<0.001 vs. IR+Mel+Sit group and $p<0.05 vs. IR+Sit group |
On the other hand, concomitant application of both melatonin and sitagliptin significantly recovered IR-induced changes in all markers of oxidative stress when compared with those of the IR group. Interestingly, the effects of the combination of melatonin and sitagliptin on MDA reduction and catalase elevation were significantly powerful than their alone administration (p<0.05) (Fig. 5a-b).
Melatonin/sitagliptin combination increased IR-induced reduction of myocardial nitric oxide in obese rats, which this effect was not reversed by 5HD: The levels of Nitric Oxide metabolites (NOx) were measured using the Griess method to quantify the myocardial NO levels in experimental groups. As shown in Fig. 6, the myocardial contents of NOx were significantly reduced in the IR group in comparison to the control group (p<0.001). Both melatonin alone and sitagliptin alone significantly recovered the changes in NOx level compared with the IR group (p<0.05). Also, combination therapy elevated the NOx level strongly (p<0.001). However, inhibition of mitochondrial K-ATP channels by 5HD was not able to suppress the effect of combination therapy on NOx level (Fig. 6), indicating that the effect of combination therapy with melatonin and sitagliptin on myocardial NO production in obese rats was not dependent on the activation of mitochondrial K-ATP channels.
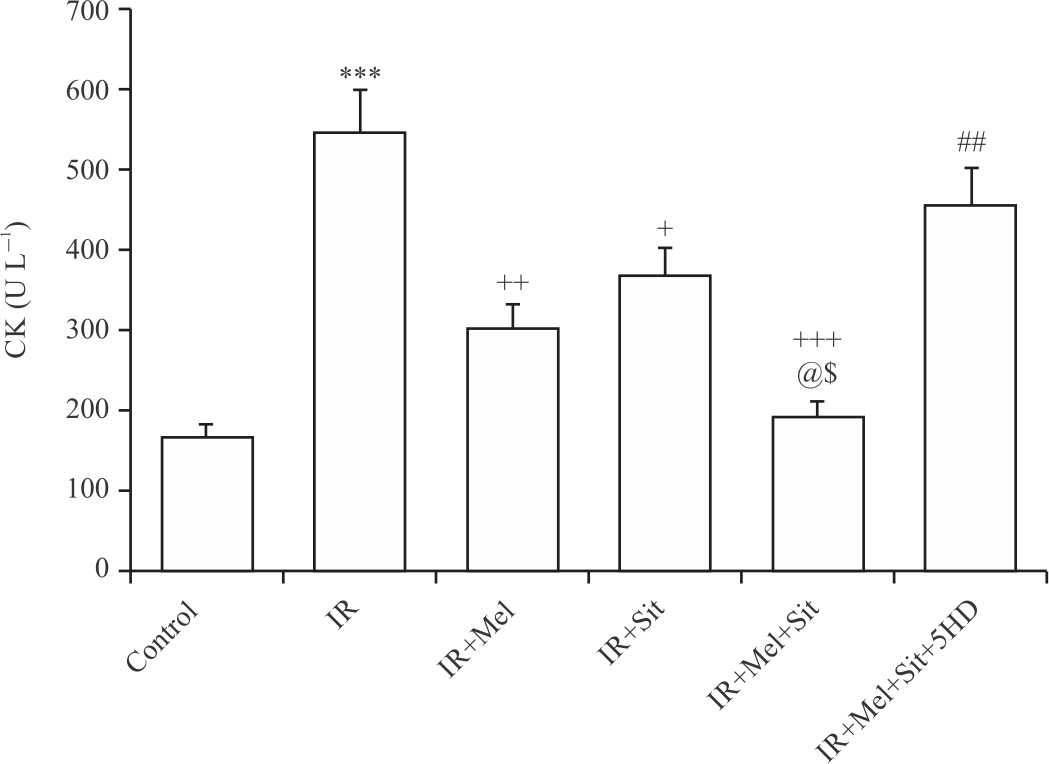 |
| Fig. 4: | CK release following myocardial IR injury in experimental groups Mean±SD and n = 6. IR: Ischemia-reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, CK: Creatine kinase, ***p<0.001 vs. control group, +p<0.05, ++p<0.01, +++p<0.001 vs. IR group, ##p<0.01 vs. IR+Mel+Sit group, @p<0.05 vs. IR+Mel group and $p<0.05 vs. IR+Sit group |
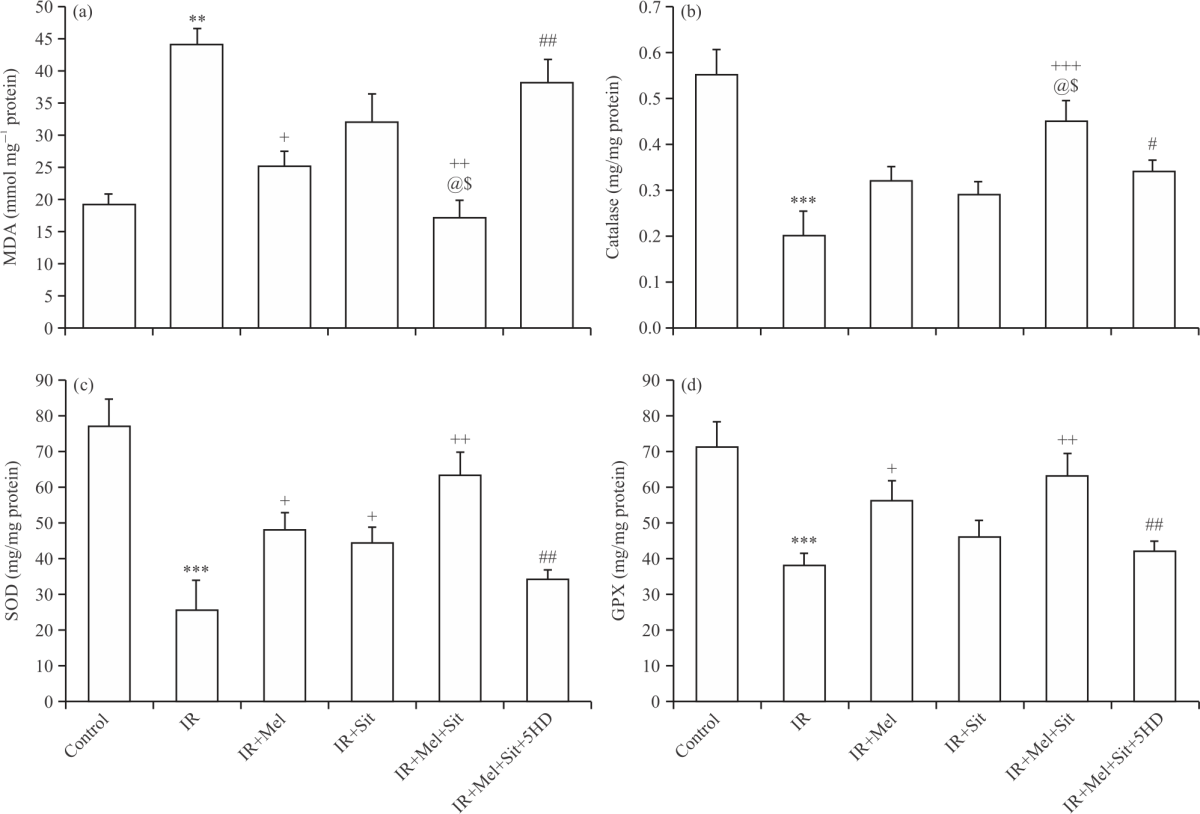 |
| Fig. 5(a-d): | Oxidative stress markers following myocardial IR injury in experimental groups, (a) Malondialdehyde (MDA), (b) Catalase, (c) Superoxide dismutase (SOD) and (d) Glutathione peroxidase (GPX) Mean±SD and n = 6. IR: Ischemia reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, **p<0.01, ***p<0.001 vs. control group, +p<0.05, ++p<0.01, +++p<0.001 vs. IR group, #p<0.05, ##p<0.01 vs. IR+Mel+Sit group, @p<0.05 vs. IR+Mel group and $p<0.05 vs. IR+Sit group |
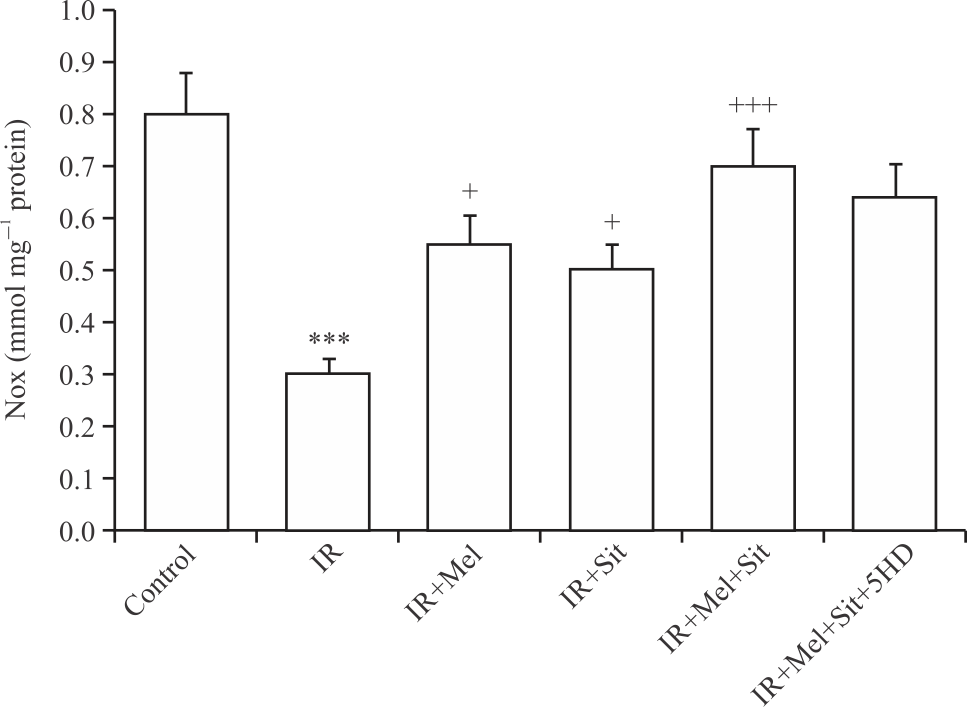 |
| Fig. 6: | Nitric oxide metabolites (NOx) following myocardial IR injury in experimental groups Mean±SD and n = 6. IR: Ischemia-reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, ***p<0.001 vs. control group, +p<0.05 and +++p<0.001 vs. IR group |
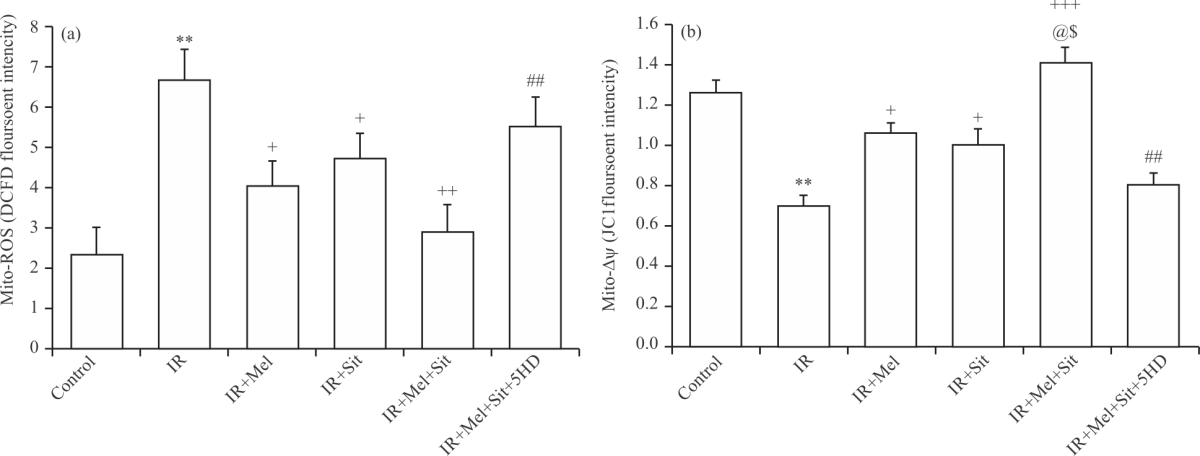 |
| Fig. 7(a-b): | Mitochondrial function markers following myocardial IR injury in experimental groups, (a) Mitochondrial reactive oxygen species (mito-ROS) and (b) Membrane changes (mito-ΔΨ) IR: Ischemia-reperfusion injury, Mel: Melatonin, Sit: Sitagliptin, 5HD: 5-hydroxydecanoic acid, Mean±SD and n = 6,**p<0.01 vs. control group, +p<0.05, ++p<0.01, +++p<0.001 vs. IR group, ##p<0.01 vs. IR+Mel+Sit group, @p<0.05 vs. IR+Mel group and $p<0.05 vs. IR+Sit group |
Melatonin/sitagliptin combination reduced IR-induced mitochondrial dysfunction in obese rats, which reversed by 5HD: Mitochondrial ROS and membrane potential change were measured to estimate the function of mitochondria. Following IR injury, the production of mitochondrial ROS (Fig. 7a) was increased and mitochondrial membrane potential (Fig. 7b) was decreased significantly in comparison with control rats (p<0.01). Administration of melatonin and/or sitagliptin, alone or in combination, significantly restored IR-induced changes in mitochondrial ROS and membrane potential. Moreover, mitochondrial membrane potential was significantly increased by combination therapy when compared with individual treatments (p<0.05) (Fig. 7b). Finally, co-administration of 5HD significantly attenuated the beneficial effects of combination therapy on mitochondrial ROS level and membrane potential change (Fig. 7).
DISCUSSION
In this study, postconditioning of obese rats with melatonin and/or sitagliptin lessened the ventricular arrhythmias induced by myocardial IR injury and this antiarrhythmic effect was associated with diminished myocardial CK release and oxidative stress and improved NO production and mitochondrial function. The results showed that the effect of the combination of melatonin/sitagliptin was greater than those of their treatments. Blockade of mitochondrial K-ATP channel via concomitant administration of its selective inhibitor 5HD abolished the beneficial and protective effects of combination therapy on all parameters except NOx levels. Therefore, the melatonin/sitagliptin combination demonstrated potent antiarrhythmic properties and reduced oxidative stress and mitochondrial function by activating the mitochondrial K-ATP channels in obese rats subjected to myocardial IR injury. Its effect on No production was not dependent on the activity of mitochondrial K-ATP channels.
The occurrence of myocardial arrhythmias following IR injury is a common outcome predisposing cell death and life-threatening consequences20. Being a main cardiovascular risk factor, obesity and weight gain also raise the risk of myocardial arrhythmias and sudden death from cardiac problems. It has been reported that increased adiposity along with subsequent metabolic complications contributes to the abnormal arrhythmogenic substrates21. Various cascades of pathological events and mediators are suggested to be crucial in IR-related cell injury and may contribute to arrhythmogenesis. These include endothelial dysfunction and NO synthase inactivation, production of pro-inflammatory cytokines and chemokines, excessive oxidative stress, mitochondrial dysfunction imbalance in ionic homeostasis and electrical activity20,22. The normal electrical activity of the heart is disturbed in the setting of hyperlipidemia and obesity due to obesity-induced metabolic disturbances as well as changes in cardiac ultrastructure, lipid peroxidation and consequent oxidative stress22,23. In our study, the induction of myocardial IR injury in obese rats showed higher degrees of myocardial arrhythmias and increased the count, timing, incidence and severity of ventricular arrhythmias PVC, VT and VF. In accompanying with these findings, NO production and antioxidative enzymes SOD, GPX and catalase were reduced and lipid peroxidation marker MDA and mitochondrial ROS were increased following IR injury.
Abrupt reperfusion in obese rats may cause ROS overproduction and severe oxidative stress damage and consequent irregular cardiac rhythm20. Insufficient NO production also exaggerates this situation24. Previous studies have reported that both melatonin and sitagliptin have considerable antioxidative effects25,26. In this regard, we showed that the antioxidative action of melatonin alone was greater than those of sitagliptin alone and that their combined action was more efficient than those of their treatments. A similar scenario was seen in the antiarrhythmic properties of these drugs. Therefore, it seems that intracellular and metabolic changes during chronic obesity may negatively interfere with the protective impacts of individual interventions23. Thus, by increasing the potency of interventions through combining them and concomitant administration, their effectiveness increases in an additive manner, leading to powerful activation of cellular pro-survival mediators, greater cardioprotection and considerable anti-arrhythmogenesis.
To explore the mechanism responsible for the antiarrhythmic and antioxidative effects of melatonin/ sitagliptin combination therapy in the hearts of obese rats, we considered the role of the mitochondrial K-ATP channels. These channels play a key role in mitochondrial homeostasis during myocardial IR injury27. Blocking these channels using concomitant administration of 5HD significantly suppressed the protective effects of melatonin/sitagliptin combination effects on ventricular arrhythmias, oxidative stress markers and mitochondrial function indicators (ROS and membrane potential). Opening of mitochondrial K-ATP channels during the reperfusion phase is associated with improved ionic homeostasis across the mitochondrial membranes, reduction of mitochondrial swelling and prevention from membrane potential collapse4,27,28. These outcomes cause the better function of the mitochondria and slow down ROS overproduction and pro-apoptotic factor release. On the other hand, the administration of 5HD could not abolish the effect of combination therapy on no production. This indicates that this effect of combination therapy in obese rats was not dependent on the activation of mitochondrial K-ATP channels and NO production by combination therapy has occurred through other mechanisms that need further investigation. Studies show that NO can trigger mitochondrial K-ATP channel opening by increasing Cyclic Guanosine Monophosphate (cGMP), leading to heart protection and reducing arrhythmias29. Hence, concomitant application of melatonin and sitagliptin additively increases NO production and activates mitochondrial K-ATP channels/mitochondrial function/ROS pathway in parallel to reduce reperfusion-induced ventricular arrhythmias in obese rats subjected to IR injury. Understanding how and in what way this combination therapy triggers the opening of mitochondrial K-ATP channels warrants additional study.
CONCLUSION
The results of this study revealed that postconditioning of obese rats with melatonin and sitagliptin has beneficial cardioprotective effects against IR-induced ventricular arrhythmias. Although their individual protective effects were non-consistent in obese rats, their combination therapy was a superior approach to reduce all ventricular arrhythmias, suppress oxidative stress, increase NO production and improve mitochondrial function. Mitochondrial K-ATP channels have a crucial contribution to the cardioprotection conferred by the melatonin/sitagliptin combination. Therefore, combination therapy is a better choice than individual interventions to protect the IR heart in obese conditions.
SIGNIFICANCE STATEMENT
This study discovers the possible additive effect of melatonin and sitagliptin combination that can be beneficial for reducing ischemia/reperfusion-induced myocardial arrhythmias in obese rats. This study will help the researcher to uncover the critical area of loss of cardioprotection in the presence of cardiovascular comorbidities that many researchers were not able to explore. Thus, a new theory on this combination therapy and possibly other combinations, may be arrived at.
REFERENCES
- Cercato, C. and F.A. Fonseca, 2019. Cardiovascular risk and obesity. Diabetology Metab. Syndr., Vol. 11.
CrossRefDirect Link - Femminò, S., P. Pagliaro and C. Penna, 2020. Obesity and cardioprotection. Curr. Med. Chem., 27: 230-239.
CrossRefDirect Link - Matsuda, M. and I. Shimomura, 2013. Increased oxidative stress in obesity: Implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis and cancer. Obesity Res. Clin. Pract., 7: e330-e341.
CrossRefDirect Link - Nakaya, H., 2014. Role of ATP-sensitive K+ channels in cardiac arrhythmias. J. Cardiovasc. Pharmacol. Ther., 19: 237-243.
CrossRefDirect Link - Lebuffe, G., P.T. Schumacker, Z.H. Shao, T. Anderson, H. Iwase and T.L.V. Hoek, 2003. ROS and NO trigger early preconditioning: Relationship to mitochondrial KATP channel. Am. J. Physiol. Heart Circulatory Physiol., 284: H299-H308.
CrossRefDirect Link - Patel, R., S. Pramanik, N.N. Rathwa, N.R. Parmar and H. Dhimmar et al., 2019. 112-LB: Melatonin and DPP-IV inhibitor: A novel combinatorial approach for ß-cell regeneration. Diabetes, Vol. 68.
CrossRefDirect Link - Forrestel, A.C., S.U. Miedlich, M. Yurcheshen, S.D. Wittlin and M.T. Sellix, 2017. Chronomedicine and type 2 diabetes: Shining some light on melatonin. Diabetologia, 60: 808-822.
CrossRefDirect Link - Jiki, Z., S. Lecour and F. Nduhirabandi, 2018. Cardiovascular benefits of dietary melatonin: A myth or a reality?. Front. Physiol., Vol. 9.
CrossRefDirect Link - Gul‐Kahraman, K., M. Yilmaz‐Bozoglan and E. Sahna, 2019. Physiological and pharmacological effects of melatonin on remote ischemic perconditioning after myocardial ischemia‐reperfusion injury in rats: Role of Cybb, Fas, NfκB, irisin signaling pathway. J. Pineal Res., Vol. 67.
CrossRefDirect Link - Waseem, M., H. Tabassum and S. Parvez, 2016. Melatonin modulates permeability transition pore and 5-hydroxydecanoate induced KATP channel inhibition in isolated brain mitochondria. Mitochondrion, 31: 1-8.
CrossRefDirect Link - El-Agamy, D.S., H.M. Abo-Haded and M.A. Elkablawy, 2016. Cardioprotective effects of sitagliptin against doxorubicin-induced cardiotoxicity in rats. Exp. Biol. Med., 241: 1577-1587.
CrossRefDirect Link - Nakamura, T., Y. Iwanaga, Y. Miyaji, R. Nohara, T. Ishimura and S. Miyazaki, 2016. Cardiovascular efficacy of sitagliptin in patients with diabetes at high risk of cardiovascular disease: A 12-month follow-up. Cardiovasc. Diabetol., Vol. 15.
CrossRefDirect Link - Li, S., H. Li, R. Wang and J.P. Zhang, 2017. The effect of sitagliptin on obese patients with insulin treatment-induced diabetes mellitus. Eur. Rev. Med. Pharmacol. Sci., 21: 3490-3495.
Direct Link - Kamble, H.V. and S.L. Bodhankar, 2014. Cardioprotective effect of concomitant administration of trigonelline and sitagliptin on cardiac biomarkers, lipid levels, electrocardiographic and heamodynamic modulation on cardiomyopathy in diabetic wistar rats. Biomed. Aging Pathol., 4: 335-342.
CrossRefDirect Link - Nauck, M.A., D.K. McGuire, K.S. Pieper, Y. Lokhnygina and T.E. Strandberg et al., 2019. Sitagliptin does not reduce the risk of cardiovascular death or hospitalization for heart failure following myocardial infarction in patients with diabetes: Observations from TECOS. Cardiovasc. Diabetol., Vol. 18.
CrossRefDirect Link - Shi, S., S. Lei, C. Tang, K. Wang and Z. Xia, 2019. Melatonin attenuates acute kidney ischemia/reperfusion injury in diabetic rats by activation of the SIRT1/Nrf2/HO-1 signaling pathway. Biosci. Rep., Vol. 39.
CrossRefDirect Link - Li, J., W. Zhou, W. Chen, H. Wang, Y. Zhang and T. Yu, 2020. Mechanism of the hypoxia inducible factor 1/hypoxic response element pathway in rat myocardial ischemia/diazoxide post‑conditioning. Mol. Med. Rep., 21: 1527-1536.
CrossRefDirect Link - Kielkopf, C.L., W. Bauer and I.L. Urbatsch, 2020. Bradford assay for determining protein concentration. Cold Spring Harbor Protoc., Vol. 2020.
CrossRefDirect Link - Sivandzade, F., A. Bhalerao and L. Cucullo, 2019. Analysis of the mitochondrial membrane potential using the cationic JC-1 dye as a sensitive fluorescent probe. Bio Protoc., Vol. 9.
CrossRefDirect Link - Yang, C.F., 2018. Clinical manifestations and basic mechanisms of myocardial ischemia/reperfusion injury. Tzu Chi Med. J., 30: 209-215.
CrossRefDirect Link - Pathak, R.K., R. Mahajan, D.H. Lau and P. Sanders, 2015. The implications of obesity for cardiac arrhythmia mechanisms and management. Can. J. Cardiol., 31: 203-210.
CrossRefDirect Link - Sedova, K.A., O.G. Bernikova, J.I. Cuprova, A.D. Ivanova and G.A. Kutaeva et al., 2019. Association between antiarrhythmic, electrophysiological and antioxidative effects of melatonin in ischemia/reperfusion. Int. J. Mol. Sci., Vol. 20.
CrossRefDirect Link - Andreadou, I., R. Schulz, L. Badimon, A. Adameová and P. Kleinbongard et al., 2020. Hyperlipidaemia and cardioprotection: Animal models for translational studies. Br. J. Pharmacol., 177: 5287-5311.
CrossRefDirect Link - Sansbury, B.E. and B.G. Hill, 2014. Regulation of obesity and insulin resistance by nitric oxide. Free Radical Biol. Med., 73: 383-399.
CrossRefDirect Link - Reiter, R.J., J.C. Mayo, D.X. Tan, R.M. Sainz, M. Alatorre-Jimenez and L. Qin, 2016. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res., 61: 253-278.
CrossRefDirect Link - Prakash, S., U. Rai, A. Uniyal, V. Tiwari and S. Singh, 2020. Sitagliptin mitigates oxidative stress and up-regulates mitochondrial biogenesis markers in brown adipose tissues of high-fat diet fed obese mice through AMPK phosphorylation. Obesity Med., Vol. 19.
CrossRefDirect Link - Kuznetsov, A.V., S. Javadov, R. Margreiter, M. Grimm, J. Hagenbuchner and M.J. Ausserlechner, 2019. The role of mitochondria in the mechanisms of cardiac ischemia-reperfusion injury. Antioxidants, Vol. 8.
CrossRefDirect Link - Oldenburg, O., M.V. Cohen, D.M. Yellon and J.M. Downey, 2002. Mitochondrial KATP channels: Role in cardioprotection. Cardiovasc. Res., 55: 429-437.
CrossRefDirect Link - Totzeck, M., U.B. Hendgen-Cotta and T. Rassaf, 2017. Nitrite-nitric oxide signaling and cardioprotection. Adv. Exp. Med. Biol., 982: 335-346.
CrossRefDirect Link








