ABSTRACT
Background and Objective: Rheumatoid Arthritis (RA) is a progressive systemic autoimmune disorder that occurs more often in females than males, being primarily found in the elderly. The present study aimed to develop and observe the effect of intraarticular injection of nanoparticulate prednisolone (PD) in the treatment of the arthritic animal model. Materials and Methods: PD loaded chitosan (CS) nanoparticles were prepared by ionotropic external gelation technique followed by the development of intraarticular injection. The PD-CS nanoparticles were evaluated for yield, drug loading, encapsulation efficiency, particle size, zeta potential, surface morphology study Polydispersity Index (PDI), in vitro release study in simulated synovial fluid, drug loading and Encapsulation Efficiency (EE) and in vivo antiarthritic activity in the carrageenan-induced rabbit model. Results: The nanoparticles were formed with a higher production yield of more than 90% in all batches. The particle size was observed between 110-198 nm with desirable zeta potential and PDI indicating excellent stability of the nanosuspensions. EE was observed between 75.89-98.95% while drug loading of 19.25-39.18%. The surface morphological study revealed the spherical, smooth and free-flowing nature of the dried nanoparticles. Optimized formulation (NP6) for 32 hrs indicating potential use in arthritic conditions. Within 1 hr, intramuscular injection of nanoparticles showed nearly 80% oedema inhibition in Group III animals while conventional injection just showed 11% inhibition. Nearly 4 fold inhibition in oedema was observed in animals injected with nanoparticulate formulation compared to conventional injection. Conclusion: PD-CS nanoparticulate IA injection would be one of the best options for the treatment of arthritis.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijp.2022.611.617
URL: https://scialert.net/abstract/?doi=ijp.2022.611.617
INTRODUCTION
Rheumatoid Arthritis (RA) is an autoimmune disease mostly found in females and elder patients. It has a global prevalence of about 1%, with women having a 2-3 times higher frequency than men1. The synovial joint lining is primarily affected in RA, which may result in progressive disease, premature death and socioeconomic pressures. Arthralgia, swelling, redness and even a loss in the range of motion are all signs of symmetrical joint involvement2. Arthritis involves the joints at the tips of the fingertips, the base of the wrist, the spine, lower back, knees and hips3. In women, RA usually begins between the ages of 30 and 60 and in men, it begins later in life. A woman's lifetime risk of having RA is 3.6%, whereas a man's lifetime risk is 1.7%. RA, on the other hand, will hit someone at any age, even small children4. Wear and tear from age and diet, illnesses, trauma, autoimmune disorders, obesity, and occupational hazards are all possible causes of arthritis5.
Nonsteroidal Anti-inflammatory Drugs (NSAIDs), steroids, disease-modifying anti-rheumatic drugs and biological response modifiers are among the available treatment options for arthritis6. The majority of medications have a variety of side effects, including GI ulcers, bleeding and irritations when taken orally. Similarly, the use of the topical and transdermal delivery technique is restricted due to low bioavailability7. To address the shortcomings of traditional drug delivery techniques, a new route of drug delivery is necessary and in this case, Intra-Articulate (IA) drug delivery is expected to provide more effective therapy for arthritis due to its potential to alleviate joint inflammation, destruction and pain while restoring patient activity levels and joint function8. The system's ability to rapidly release drug material to the infected joint region means that high drug concentrations can be delivered with little or no systemic toxicity9,10.
Since most drugs are quickly removed from the joint following a conventional injection. Because of the short course of action, repeated injections are required, resulting in high medication rates, low patient compliance and injection-related complications (e.g., infection, post-injection flare, crystal-induced synovitis, cutaneous atrophy). The most challenging part of designing an intraarticular drug delivery system is increasing the therapeutic agent's survival time in the joint. Many different types of particulate carriers have been studied to extend the retention time of the therapeutic agents in the knee cavity. Due to promising prolonged and extended drug release properties in joints, higher surface area and nanoscale dimensions, micro and nanoparticulate drug delivery systems (microspheres, liposomes, nanosponges) have drawn the interest of scientists in recent years. Furthermore, these innovations assist in drug retention, decreased drug clearance from joints and increased therapeutic potential and patient compliance11. In contrast to traditional rapid injections, an IA injection paired with prolonged or sustained release nanoparticles would be a potential alternative therapy for arthritis. Shi-Yin Zhou et al.8 developed intra-articular injection of dexamethasone microparticles and optimized formulation showed an excellent antiarthritic effect in the carrageenan-induced arthritic rabbit model after intraarticular injection. Similarly, Rabab Kamel et al.12 developed PLA coated co-micellar nanosystem of resveratrol as an intra-articular injection for the treatment of arthritis and found the recovery of the joints and synovial structure12.
Prednisolone (PD) is a glucocorticoid that inhibits the release of pro-inflammatory cytokines, making it an important treatment for rheumatoid arthritis. It is mostly used to treat multiple sclerosis, rheumatoid arthritis, autoimmune hepatitis and other inflammatory and autoimmune disorders13. Since it inhibits gene transcription for COX-2, cytokines, cell adhesion molecules and inducible NO synthetase, PD is also known as a "disease-modifying antiarthritic drug." It induces systemic adverse effects such as bone destruction, increased susceptibility to illness, osteoporosis, peptic ulcers and buffalo hump when administered orally14. The parenteral route of administration results in a high degree of drug clearance, which necessitates invasive and repeated drug administration15.
Considering all these potential drawbacks of the current drug delivery technologies, our research group has attempted to develop an alternate delivery system in intraarticular injection form containing PD nanoparticles for efficient treatment of arthritis in the animal model.
MATERIALS AND METHODS
Study area: The study was carried out at the Department of Traditional Chinese Medicine, The Fourth Hospital of Baotou, Baotou City, Inner Mongolia, 014030, China from January, 2020-April, 2021.
Materials: PD was obtained as a gift sample from Zhejiang Hisun Pharmaceutical Company (Taizhou City, China), Chitosan (Deacetylationdegree of 70-85%)was purchased from Weifang Yuexiang Chemical Co. Ltd. China. Tripolyphosphate (TPP), Tween 80, Acetic acid and Trehalose were purchased from Sigma Aldrich China. Methanol, Dichloromethane (DCM) and other solvents and reagents were purchased from Merck, Germany. All other analytical grade reagents were used during the experiments.
| Table 1: | Formulation batches of PD-CS nanoparticles | |||||
| Batch | NP1 | NP2 | NP3 | NP4 | NP5 | NP6 |
| PD (mg) | 10 | 10 | 10 | 10 | 10 | 10 |
| Methanol (mL) | 10 | 10 | 10 | 10 | 10 | 10 |
| Chitosan (mg) | 30 | 60 | 90 | 120 | 150 | 180 |
| 3% acetic acid (mL) | 15 | 15 | 15 | 15 | 15 | 15 |
| Tween 80 (mL) | 0.1 | 0.2 | 0.3 | 0.4 | 0.5 | 0.6 |
| TPP (mg) | 5 | 5.5 | 6 | 6.5 | 7 | 7.5 |
| Purified water | q.s. | q.s. | q.s. | q.s. | q.s. | q.s. |
| NP: Optimized formulation, TPP: Tripolyphosphate and PD: Prednisolone | ||||||
Preparation of CS-PD nanoparticles: PD loaded CS nanoparticles were prepared by ionotropic external gelation technique using TPP as a counterion16. CS (30-180 mg) was dissolved in 3% v/v of acetic acid solution (15 mL) under continuous stirring to get a slightly viscous solution. Tween 80 (0.1-0.6 mL) was added to this polymeric solution and stirred for 30-45 min (Table 1). PD (10 mg) was dissolved at a concentration of 1 mg mL–1 in methanol in a test tube using the ultrasonication technique. The drug solution was added dropwise to the polymeric solution using a syringe fitted with a 19 gauge needle. These 2 phases were allowed to mix properly for 30-45 min on a magnetic stirrer. TPP (5-7.5 mg) was dissolved separately in double-distilled water and added dropwise to drug-polymer dispersion under continuous magnetic stirring for 12-15 hrs to complete the crosslinking between CS and TPP. The nanoparticulate suspension thus obtained was centrifuged at -50-70°C using a cooling ultracentrifuge at 30,000 RPM. Nanoparticles were separated and the supernatant liquid was used for the determination of unentrapped PD from nanoparticles. Further, PD-loaded CS nanoparticles were lyophilized using Trehalose as a cryoprotectant to get free-flowing powder.
Preparation of intraarticular injection: PD nanoparticles equivalent to 10 mg were suspended in water for injection followed by ultrasonication for 15-20 min. Sodium chloride equivalent to 0.9% and HPMC 0.1% was added to this suspension under continuous stirring till gets completely dissolved. The whole system was kept in the refrigerator at 2-8 for 24 hrs. This suspension was filled in clear transparent vials and sterilized by autoclaving at 121 at 15 psi for 20 min.
Evaluation of PD-CS nanoparticles
Yield of PD-CS nanoparticles: The percentage yield of nanoparticles was determined for each batch using the following formula17:
 |
Encapsulation efficiency (EE) of PD: The nanoparticulate suspension obtained during experiments was centrifuged at -50-70°C using a cooling ultracentrifuge at 30,000 RPM. The supernatant solution thus obtained was diluted 10 times using double distilled water and analyzed by HPLC with a UV detector at 260 nm. EE of PD nanoparticles was determined by using the following formula17:
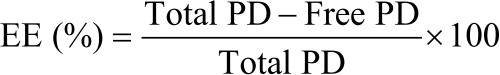 |
PD loading in CS nanoparticles (DL): PD nanoparticles equivalent to 10 mg were dissolved in 3% acetic acid solution ( 20 mL) under continuous magnetic stirring till nanoparticles get completely dissolved followed by the addition of methanol (15 mL) to extract the PD. The resulted solution was filtered through a 0.3-micron disk filter to get a clear solution. This solution was analyzed by HPLC with a UV detector at 260 nm. Drug Loading (DL) was determined by using the following formula17:
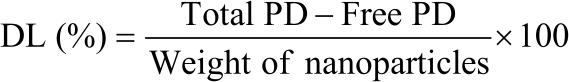 |
In vitro PD release study: The PD release from CS nanoparticles was performed using a dialysis membrane (molecular weight cut of 10 KDa) in simulated synovial fluid18. Exactly 10 mg equivalent PD nanoparticles were dispersed in 10 mL simulated synovial fluid and poured in dialysis tube already tied from end using thread. Another end of the tube was also tied properly to avoid the leakage of internal fluid containing nanoparticles. This tubing was then placed in the glass beaker containing 200 mL simulated synovial fluid maintained at 37 on a magnetic stirrer with continuous stirring at 50 RPM. The samples (0.5 mL) were withdrawn at the predetermined time interval and replenished with an equal volume of synovial fluid to maintain the sink condition. PD content released at each time point was determined at 260 nm by injecting samples to HPLC.
Surface morphology study: The surface characteristics of the developed nanoparticles were studied using scanning electron microscopy (FESEM-S 4800, Hitachi, Japan). The shape, size and, surface morphology were observed at different locations.
Particle size, zeta potential and polydispersity index (PDI): Diluted (1:10) and properly sonicated sample of nanoparticles loaded in malvern particle size analyzer (Mastersizer 3000) to determine the particle size distribution, zeta potential and PDI of the suspensions.
Efficacy in the carrageenan-induced arthritis rabbit model: All animal handlings and experiments were performed with prior approval of the institutional animal ethical committee of the Department of Traditional Chinese Medicine, The Fourth Hospital of Baotou, Baotou City, Inner Mongolia, 014030, China. New Zealand white male and female rabbits (n = 30) weighing 2.1-2.9 kg were divided into 3 groups each containing 10 animals and housed in cages for one week before actual experiments. Carrageenan suspension (15%) prepared in pyrogen-free saline buffer was injected (0.2 mL) in all animals to induce paw oedema. The initial volume of oedema was determined after 24 hrs of carrageenan injection using the micrometre. Group I animals was not given any treatment so considered as a control animal group while Group II animals were treated with an intramuscular conventional injection of PD. Group III animals were treated with an intramuscular injection of PD containing nanoparticles (Optimised formulation). The swelling of the paw was determined at a predetermined time interval of 48 hrs. The antiarthritic potential was determined by calculating the % inhibition of oedema against the control group19.
RESULTS AND DISCUSSION
PD loaded chitosan nanoparticles prepared by ionotropic external gelation technique showed the following physicho-chemical characteristics as mentioned in Table 2.
The nanoparticles were formed with a higher production yield of more than 90% in all batches, indicating minimal loss of excipients and other nanoparticle components. In the presence of surfactant, this finding confirms full cross-linking between the CS (positive charge) and TPP (negative charge). With the CS polymer concentration, particle size increased (110-198 nm). Based on the zeta potential values, the PDI values (less than 0.3) of the batches NP2, NP5 and NP6 indicated the monodispersed existence of the nanosuspensions with excellent stability. The zeta potential between -30±30 mV, preserves the proper repulsive force and increases the physical integrity of nanoparticulate dispersion or suspension20. Drug loading and EE values were also found higher indicating the acceptability of the nanoparticles. The surface morphological study revealed the spherical, smooth and free-flowing nature of the dried nanoparticles as shown in Fig. 1.
In vitro, PD release from nanoparticle formulations was performed in a simulated synovial fluid along with pure PD. All nanoparticle batches showed excellent sustained release behaviour ranging from 12-32 hrs as shown in Fig. 2. Such sustained and controlled release behaviour would be useful in the treatment of arthritis. This delivery technology would bring a reduction in dosing frequency as compared to conventional injectable systems. Shi-Yin Zhou et al.8 developed dexamethasone intra-articular injection containing microspheres and found drug release for 25 days. Similarly, Dong et al.21 also developed a Novel intraarticular injection of diclofenac for the treatment of arthritis and optimized formulation showed initial burst release followed by a sustained release for 12 hrs. A similar type of release pattern was also observed in the case of our optimized formulation (NP6) for 32 hrs indicating potential use in arthritic conditions.
| Table 2: | Physicho-chemical characteristics of PD-CS nanoparticles | |||||
| Batch code | NP1 | NP2 | NP3 | NP4 | NP5 | NP6 |
| Yield (%) | 92.11 | 93.14 | 89.25 | 94.15 | 95.16 | 93.56 |
| Particle size (nm) | 110 | 124 | 135 | 149 | 165 | 198 |
| PDI | 0.35 | 0.29 | 0.45 | 0.31 | 0.25 | 0.15 |
| Zeta potential (mV) | 18.14 | 21.36 | 25.63 | 23.28 | 22.11 | 20.12 |
| Drug loading (%) | 19.25 | 25.25 | 30.51 | 32.50 | 35.40 | 39.18 |
| Encapsulation efficiency (%) | 75.89 | 89.58 | 91.20 | 93.21 | 95.20 | 98.95 |
| NP: Optimized formulation and PDI: Polydispersity index | ||||||
| Table 3: | Inhibition of (%) oedema in animal groups | ||||
| Inhibition of (%) oedema (hrs) | |||||
| Groups | 1 | 2 | 4 | 12 | 24 |
| I (Control) | No oedema inhibition | ||||
| II (Conventional injection) | 10.15 | 17.40 | 21.24 | 25.12 | 29.45 |
| III (PD-CS nanoparticles injection) | 78.90 | 89.45 | 95.24 | 100 | 100 |
| PD-CS: Prednisolone loaded chitosan | |||||
 |
| Fig. 1: | Scanning electron microscopy of PD loaded CS nanoparticles showing spherical surface characteristics |
 |
| Fig. 2: | Comparative PD release from nanoparticle batches showing excellent sustained release behaviour |
The antiarthritic activity of the intramuscular injection of PD-CS nanoparticles was determined in the carrageenan-induced arthritis model. In this research study, intramuscular injection of PD-CS nanoparticles showed the greatest anti-inflammatory activity through oedema inhibition. Within 1 hr, intramuscular injection of nanoparticles showed nearly 80% oedema inhibition in Group III animals (Table 3) while conventional injection just showed 11% inhibition.
 |
| Fig. 3: | Comparative (%) oedema inhibition of different formulations PD-CS NP intramuscular injection showed maximum inhibition with p<0.02 |
The complete recovery from oedema and inflammation was observed within 12 hrs in Group III animals indicating 100% oedema inhibition a shown in Fig. 3. Nearly 4 fold inhibition in oedema was observed in animals injected with nanoparticulate formulation compared to conventional injection. These results were found to be statistically significant with p<0.02. The animals treated with conventional injection showed only 30% oedema inhibition over 24 hrs time frame. These observations showed the great potential of nanoparticles in the treatment of arthritis. Similar observations were also reported by Shi-Yin Zhou et al.8 and concluded the potential use of dexamethasone intra-articular injection containing microspheres in carrageenan-induced animal models. Similarly, Dong et al.21 also observed similar outcomes after intramuscular injection of nanoparticulate diclofenac in the arthritis model.
The current research work deals with the development of PD-loaded chitosan nanoparticles followed by intramuscular injection for efficient treatment of arthritis in the animal model. The conventional injectable systems get rapidly removed from the joints which required frequent administration of medications leading to discomfort to the patients22. Oral drug delivery technologies failed due to the various side effects associated with the GI tract including irritation and bleeding7. Several previous studies have shown that chitosan has a chondroprotective effect, enhancing chondrocyte proliferation, increasing cartilage matrix component expression levels and inhibiting inflammatory and catabolic mediators after IA injection in arthritic models. As a result, chitosan is a promising polymer for the production of scaffolds and hydrogels for cartilage lesion repair, cell transplantation, long-term drug release and viscosupplementation23,24. Similarly, PD-CS nanoparticulate IA injection would be one of the best options for the treatment of arthritis.
The currently developed prednisolone loaded nanoparticulate injection would be a promising alternative approach in comparison to the conventional drug delivery system for the treatment of arthritis. The conventional gelling systems in the form of injections get easily removed from the joints and requires the repeated administration of the dose this major limitation of the conventional system could be avoided with this newly developed technique. Further applications need to be studied at the clinical level to establish the safety and efficacy of the formulation. This clinical study was not performed in the present research study and is considered as the limitation of the study.
CONCLUSION
In the present study, PD-CS nanoparticles were successfully developed and utilized in intraarticular injection. The nanoparticles served as an excellent sustained release carrier which is quite essential for arthritis treatment. Nearly 4 fold inhibition in oedema was observed in animals injected with nanoparticulate formulation compared to conventional injection indicating the best suitable option for the treatment of arthritis.
SIGNIFICANCE STATEMENT
The aforementioned investigation is directed towards to development and observation of the effect of intra-articular injection of nanoparticulate prednisolone (PD) in the treatment of the arthritic animal model. The study will propose a new method to treat arthritic patients in a fast and more effective way.
REFERENCES
- Guo, Q., Y. Wang, D. Xu, J. Nossent, N.J. Pavlos and J. Xu, 2018. Rheumatoid arthritis: Pathological mechanisms and modern pharmacologic therapies. Bone Res., Vol. 6.
CrossRefDirect Link - Kiruthika, R. and I. Ramkumar, 2020. Clinical evaluation of meghasaanthi chooranam in the treatment of uthiravaathasuronitham (Rheumatoid arthritis) - single case study. J. Res. Biomed. Sci., 3: 82-89.
CrossRefDirect Link - Grijalva, C.G., L. Kaltenbach, P.G. Arbogast, E.F. Mitchel and M.R. Griffin, 2010. Initiation of rheumatoid arthritis treatments and the risk of serious infections. Rheumatology, 49: 82-90.
CrossRefDirect Link - Metter, E.J., R. Conwit, J. Tobin and J.L. Fozard, 1997. Age-associated loss of power and strength in the upper extremities in women and men. J. Gerontol. Ser. A: Biol. Sci. Med. Sci., 52A: B267-B276.
CrossRefDirect Link - Li, C., T. Liu, W. Sun, L. Wu and Z.Y. Zou, 2015. Prevalence and risk factors of arthritis in a middle-aged and older Chinese population: The China health and retirement longitudinal study. Rheumatology, 54: 697-706.
CrossRefDirect Link - Aletaha, D. and J.S. Smolen, 2018. Diagnosis and management of rheumatoid arthritis. JAMA, 320: 1360-1372.
CrossRefDirect Link - Wen, H., H. Jung and X. Li, 2015. Drug delivery approaches in addressing clinical pharmacology-related issues: Opportunities and challenges. AAPS J., 17: 1327-1340.
CrossRefDirect Link - He, G.Q., G.W. Hu, Y.C. Wang and S.Y. Zhou, 2016. Intra-articular injection of dexamethasone microparticles: Assessment in carrageenan induced arthritic rabbit with statistical evaluation using full factorial design. J. Biomater. Tissue Eng., 6: 910-918.
CrossRefDirect Link - Son, A.R., D.Y. Kim, S.H. Park, J.Y. Jang and K. Kim et al., 2015. Direct chemotherapeutic dual drug delivery through intra-articular injection for synergistic enhancement of rheumatoid arthritis treatment. Sci. Rep., Vol. 5.
CrossRefDirect Link - Yin, N., X. Guo, R. Sun, H. Liu and L. Tang et al., 2020. Intra-articular injection of indomethacin-methotrexate in situ hydrogel for the synergistic treatment of rheumatoid arthritis. J. Mater. Chem. B, 8: 993-1007.
CrossRefDirect Link - Oliveira, I.M., C. Gonçalves, R.L. Reis and J.M. Oliveira, 2018. Engineering nanoparticles for targeting rheumatoid arthritis: Past, present and future trends. Nano Res., 11: 4489-4506.
CrossRefDirect Link - Kamel, R., H. Abbas and N.M. Shaffie, 2019. Development and evaluation of PLA-coated co-micellar nanosystem of resveratrol for the intra-articular treatment of arthritis. Int. J. Pharmaceutics, Vol. 569.
CrossRefDirect Link - Yan, F., H. Li, Z. Zhong, M. Zhou, Y. Lin, C. Tang and C. Li, 2019. Co-delivery of prednisolone and curcumin in human serum albumin nanoparticles for effective treatment of rheumatoid arthritis. Int. J. Nanomed., 14: 9113-9125.
CrossRefDirect Link - Hwang, J., K. Rodgers, J.C. Oliver and T. Schluep, 2008. α-methylprednisolone conjugated cyclodextrin polymer-based nanoparticles for rheumatoid arthritis therapy. Int. J. Nanomed., 3: 359-371.
CrossRefDirect Link - Potočnjak, I., J. Marinić, L. Batičić, L. Šimić, D. Broznić and R. Domitrović, 2020. Aucubin administered by either oral or parenteral route protects against cisplatin-induced acute kidney injury in mice. Food Chem. Toxicol., Vol. 142.
CrossRefDirect Link - Elzatahry, A.A. and M.S.M. Eldin, 2008. Preparation and characterization of metronidazole-loaded chitosan nanoparticles for drug delivery application. Polym. Adv. Technol., 19: 1787-1791.
CrossRefDirect Link - Kurakula, M., N.R. Naveen, B. Patel, R. Manne and D.B. Patel, 2021. Preparation, optimization and evaluation of chitosan-based avanafil nanocomplex utilizing antioxidants for enhanced neuroprotective effect on PC12 cells. Gels, Vol. 7.
CrossRefDirect Link - Thing, M., N. Mertz, L. Ågårdh, S.W. Larsen, J. Østergaard and C. Larsen, 2019. Simulated synovial fluids for in vitro drug and prodrug release testing of depot injectables intended for joint injection. J. Drug Delivery Sci. Technol., 49: 169-176.
CrossRefDirect Link - Achari, Y., C.R. Reno, C.B. Frank and D.A. Hart, 2012. Carrageenan-induced transient inflammation in a rabbit knee model: Molecular changes consistent with an early osteoarthritis phenotype. Inflammation Res., 61: 907-914.
CrossRefDirect Link - Tantra, R., P. Schulze and P. Quincey, 2010. Effect of nanoparticle concentration on zeta-potential measurement results and reproducibility. Particuology, 8: 279-285.
CrossRefDirect Link - Dong, W., H. Yang, M. Cheng, X. Zhang, J. Yin, Z. Zeng and G. Huang, 2020. Dexmedetomidine alleviates pulmonary ischemia-reperfusion injury through modulating the miR-21-5p/Nr4a1 signaling pathway. Acta Biochim. Pol., 67: 521-529.
CrossRefDirect Link - Maudens, P., O. Jordan and E. Allémann, 2018. Recent advances in intra-articular drug delivery systems for osteoarthritis therapy. Drug Discovery Today, 23: 1761-1775.
CrossRefDirect Link - Comblain, F., G. Rocasalbas, S. Gauthier and Y. Henrotin, 2017. Chitosan: A promising polymer for cartilage repair and viscosupplementation. Bio-Med. Mater. Eng., 28: S209-S215.
CrossRefDirect Link - Mi, F.L., Y.C. Tan, H.F. Liang and H.W. Sung, 2002. In vivo biocompatibility and degradability of a novel injectable-chitosan-based implant. Biomaterials, 23: 181-191.
CrossRefDirect Link








