Research Article
An in vitro Study: Inhibitory Effect of Carfilzomib on Human Serum Paraoxonase-1 (hPON1)
Department of Primary Education, Faculty of Education, Bursa Uludağ University, Bursa, Turkey
LiveDNA: 90.36562
Multiple Myeloma (MM) is a clonal plasma cell malignancy that accounts for approximately 13% of haematological cancers1. Multiple myeloma results from the oncogenic transformation of the white blood cells responsible for producing antibodies2. Multiple myeloma cells accumulate in the bone marrow, where the production of blood cells occurs3. The progression of multiple myeloma is assessed by measuring the amount of M protein in the serum4. Carfilzomib was approved in 2012 by the US Food and Drug Administration as a proteasome inhibitor for the treatment of multiple myeloma5. Paraoxonase (hPON1) activity was lower in multiple myeloma patient serum6. In addition, decreased activity of hPON1 has been suggested as a poor prognostic marker of multiple myeloma patients7. Human serum paraoxonase-1 (EC 3.1.8.1, hPON1) is a Ca2+ dependent lactonase associated with High-Density Lipoprotein (HDL)8. hPON1 is synthesized in the liver and is secreted into the blood. It is a glycoprote in with a molecular weight of 43-45 kDa. hPON1 has been detected in a wide variety of mammalian tissues, including human serum, by biochemical studies. However, it is not found in the blood of birds, fish and most reptiles9. hPON1 is also associated with HDL that contains Apo A1 and clusterin. In ultracentrifugation, the majority of hPON1 is found in HDL10.
hPON1 is the first discovered member of the multiple gene families, including PON1, PON2 and PON3. Three-member genes of the family are widely expressed in mammalian tissues11. hPON1 is mostly found in HDL-associated plasma, while PON2 and PON3 are not found in plasma12. hPON1 delays Low-Density Lipoprotein (LDL) oxidative modification and is therefore admitted antiatherogenic13. In addition, hPON1 is accepted to be an important factor in the antioxidant activity of HDL as it contributes to the antioxidant effect of HDL14.
In the serum of patients affected by different types of cancer, such as lung cancer, gastrointestinal cancer, breast and gynaecological cancer, prostate, bladder cancer, central nervous system tumours, non-Hodgkin lymphoma and Acute Lymphoblastic Lymphoma (ALL), hPON1 activity is significantly lower. A decrease in hPON1 activity may negatively affect the antioxidant role of the enzyme and expose it to higher oxidative stress15.
The effect of anti-cancer drugs on hPON1 has not been extensively studied. Rats treated with cyclophosphamide, a chemotherapeutic drug, showed a 2 fold increase in renal hPON1 activity. In contrast, a reduced hPON1 activity was observed in vitro during incubations with chemotherapeutic agents (cetuximab, paclitaxel, etoposide, docetaxel and ifosfamide) when purified hPON1 was used16. Oxidative stress and inflammation are considered important factors that play a role in the development of cancer and in determining the prognosis of chemotherapy. There is an inverse proportion between hPON1 enzyme activity and oxidative stress in cancer patients. If any drug reduces PON1 enzyme activity, it can occur many vascular diseases, including atherosclerosis, due to increased oxidative stress17.
In this study, we investigated the in vitro inhibition effect of carfilzomib, which is widely used in cancer treatment, on hPON1 enzyme activity.
Study area: The study was carried out at the Department of Chemistry, Research Lab, Turkey from April-July, 2021.
Chemicals: All chemicals were obtained from Sigma-Aldrich Chemie GmbH (Taufkirchen, Germany). Carfilzomib was obtained from Bursa Uludağ University Faculty of Medicine Oncology Department.
Paraoxonase activity measurement: Human serum samples were obtained from Erzincan Mengücekgazi Research Hospital. Paraoxonase activity of the enzyme was determined by using paraoxon as substrate in a 50 mM glycine/NaOH (pH: 10.5) buffer solution containing 1 mM CaCl2 at 25°C. The activity measurement is based on the absorption at 412 nm of paranitrophenol formed as a result of the reaction of paraoxon and PON1 enzyme. The enzyme unit of paraoxonase is the number of micromoles of paraoxon hydrolyzed in 1 min18. Activity measurement was performed using a spectrophotometer (Chebios UV-VIS).
Determination of IC50 and Ki constants for chemotherapeutic drug: The inhibitory effect of carfilzomib, which is widely used in cancer chemotherapy, was investigated. This chemotherapeutic drug was tested 3 times for each concentration. Paraoxonase activities of the enzyme were analyzed at different drug concentrations. Graphs were plotted showing percent activity for carfilzomib as a function of drug concentration. Control activity in the absence of inhibitor was accepted as 100%. The 50% inhibition (IC50 value) of carfilzomib was obtained from the graphs using different inhibitor concentration values. To calculate Ki values, 3 different inhibitory concentrations of carfilzomib were tested Lineweaver-Burk curves were used to determine the values of Ki and the type of inhibition.
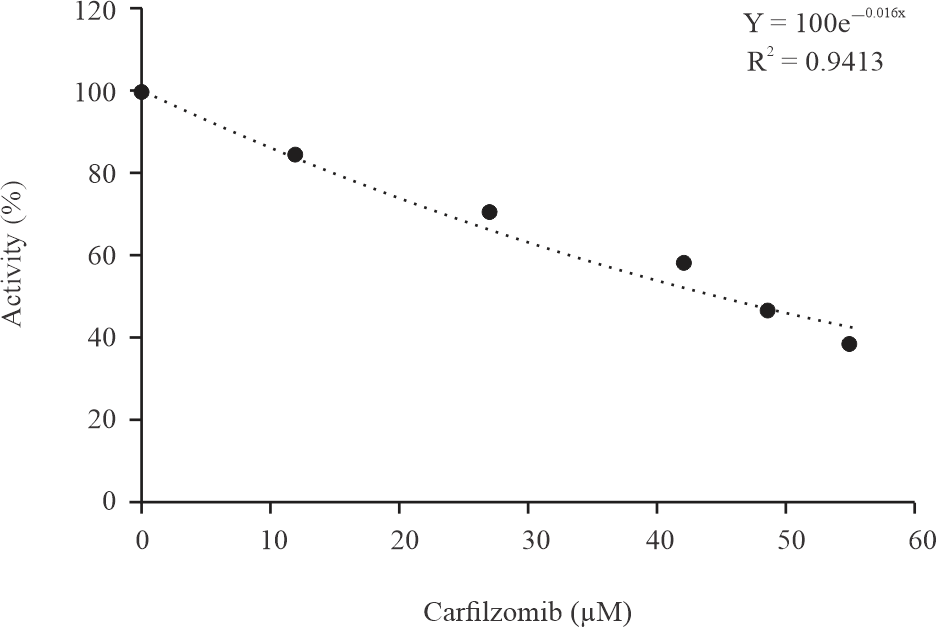 |
| Fig. 1: | Activity (%) concentration graph used to determine the IC50 value |
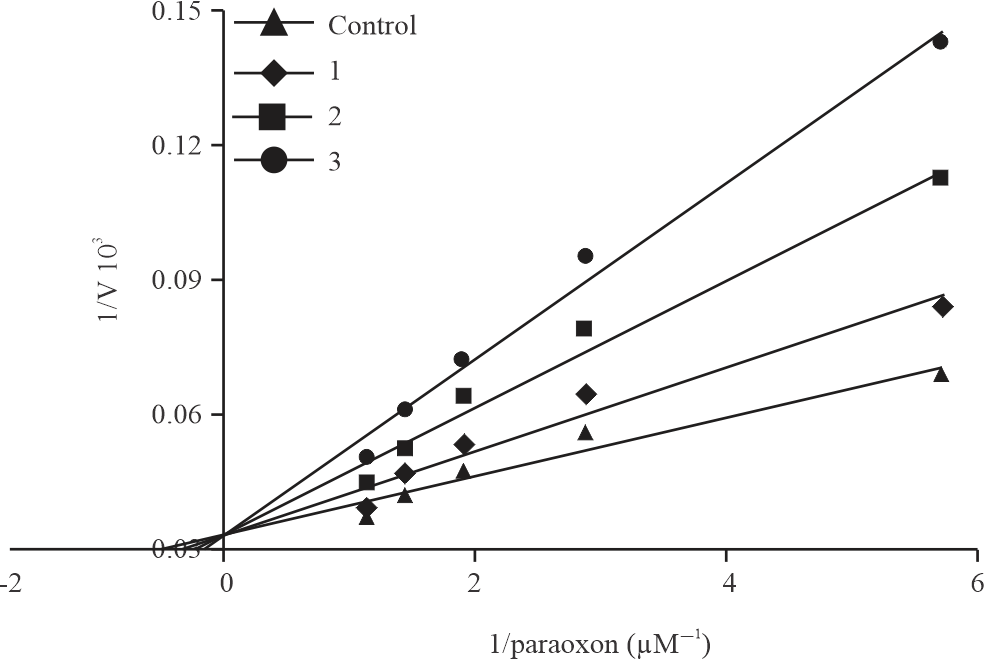 |
| Fig. 2: | Lineweaver-Burk graph used to determine Ki constant |
In this study, the in vitro inhibition effect of carfilzomib on paraoxonase enzyme activity was studied. For carfilzomib, which inhibits hPON1-IC50 value (inhibitor concentration that halves the enzyme's activity) was determined by plotting (%) activity-[Carfilzomib] graphs using 5 different inhibitor concentrations at constant paraoxon concentration. The IC50 value was calculated as 43.31 μM from the curve equation in the graph (Fig. 1).
Then, 1/V-1/paraoxon values were found by using the activity values obtained at 3 different inhibitor (I1:20, I2:40 and I3:60 μM) concentrations and 5 different paraoxon (1.33, 1.66, 2.22, 3.33 and 6.66 μM–1) concentrations for carfilzomib. Ki values were calculated by drawing lineweaver-burk graphs. The average Ki value was found to be 44.37 μM (Fig. 2). The type of inhibition is competitive.
In this study, the inhibition effect of carfilzomib, an anti-cancer drug, on paraoxonase activity was performed. The IC50 value for carfilzomib was determined as 43.31 μM using the Activity-[1] (%) graph (Fig. 1). In our study, Lineweaver-Burk graphs were used to determine the Ki constant for carfilzomib, which has an inhibitory effect on the human serum hPON1 enzyme. The Ki constant was determined as 44.37 μM (Fig. 2). The type of inhibition is competitive. Reducing the paraoxonase activity of drugs can lead to critical consequences such as cardiovascular diseases. hPON1 is one of the antioxidant defense mechanisms in the human body. There are many scavenging systems for reactive oxygen species in the human body. hPON1 protects LDL, HDL and macrophages against oxidative stress by scavenging reactive oxygen species in living metabolism. Therefore, hPON1 prevents cardiovascular diseases19. hPON1 is a calcium-dependent esterase that hydrolyzes compounds such as organophosphate tryters, aryl esters, cyclic carbamates, glucuronides, estrogen esters and thiolactones. However, the physiological substrate of hPON1 is lactones20.
There are many studies on the interactions between hPON1 activity and drugs. Statins are a group of lactone substrates. They act by reducing LDL. Decreased LDL causes an increase in hPON1 activity. Increasing hPON1 activity contributes to the reduction of oxidative stress and the prevention of cardiovascular diseases. Pravastatin, simvastatin and atorvastatin have a positive effect on hPON1 activity21-23. Aspirin is widely used in the treatment and prevention of vascular diseases. It was tested that aspirin may have beneficial effects on hPON1 activity. Aspirin use significantly increased hPON1 activity in patients with coronary artery disease24. Valsartan and barnidipine were found to not affect hPON1 activity25,26. In another study, gentamicin sulfate and cefazolin sodium decreased hPON1 activity27.
Various enzyme-anticancer drug interaction studies have been carried out in our laboratory. For example, Türkeş et al.28 examined the in vitro effects of some anti-cancer drugs, (1) Palonosetron hydrochloride, (2) Bevacizumab and (3) Cyclophosphamide on hPON1. Anti-cancer drugs must be potent inhibitors of human serum hPON1. Compared with other anti-cancer drugs, palonosetron hydrochloride was found to inhibit enzyme activity significantly. The inhibition order of drugs was determined as 1>2>3. As stated above, carfilzomib strongly inhibited the enzyme in this study. Cardiovascular disorders may occur in cancer patients using this drug, as the activity of the PON1 enzyme will decrease. However, this study can be illuminated by oncological studies.
In conclusion, we investigated the in vitro effects of carfilzomib on hPON1. We did not encounter any literature on the relationship between hPON1 and carfilzomib. However, it is known that the expression and activity of the paraoxonase enzyme are critical for cancer diseases. Many studies have shown a decrease in hPON1 activities in different cancer patients. hPON1 is a multifunctional enzyme involved in the regulation of antioxidant defence and cell behaviour. In addition, hPON1 activity protects against cardiovascular diseases. Carfilzomib is used as a chemotherapeutic drug in the treatment of cancer. When applied in cancer therapy, they may cause some metabolic disorders, especially in patients with atherosclerotic lesions. However, our results should be confirmed by some in vivo studies.
This study discovered the inhibitory effect of carfilzomib on hPON1, which many researchers failed to discover, which may be useful for understanding the adverse effects of carfilzomib on cardiovascular diseases. Thus, a new theory may have been reached regarding the use of carfilzomib in cancer treatment.