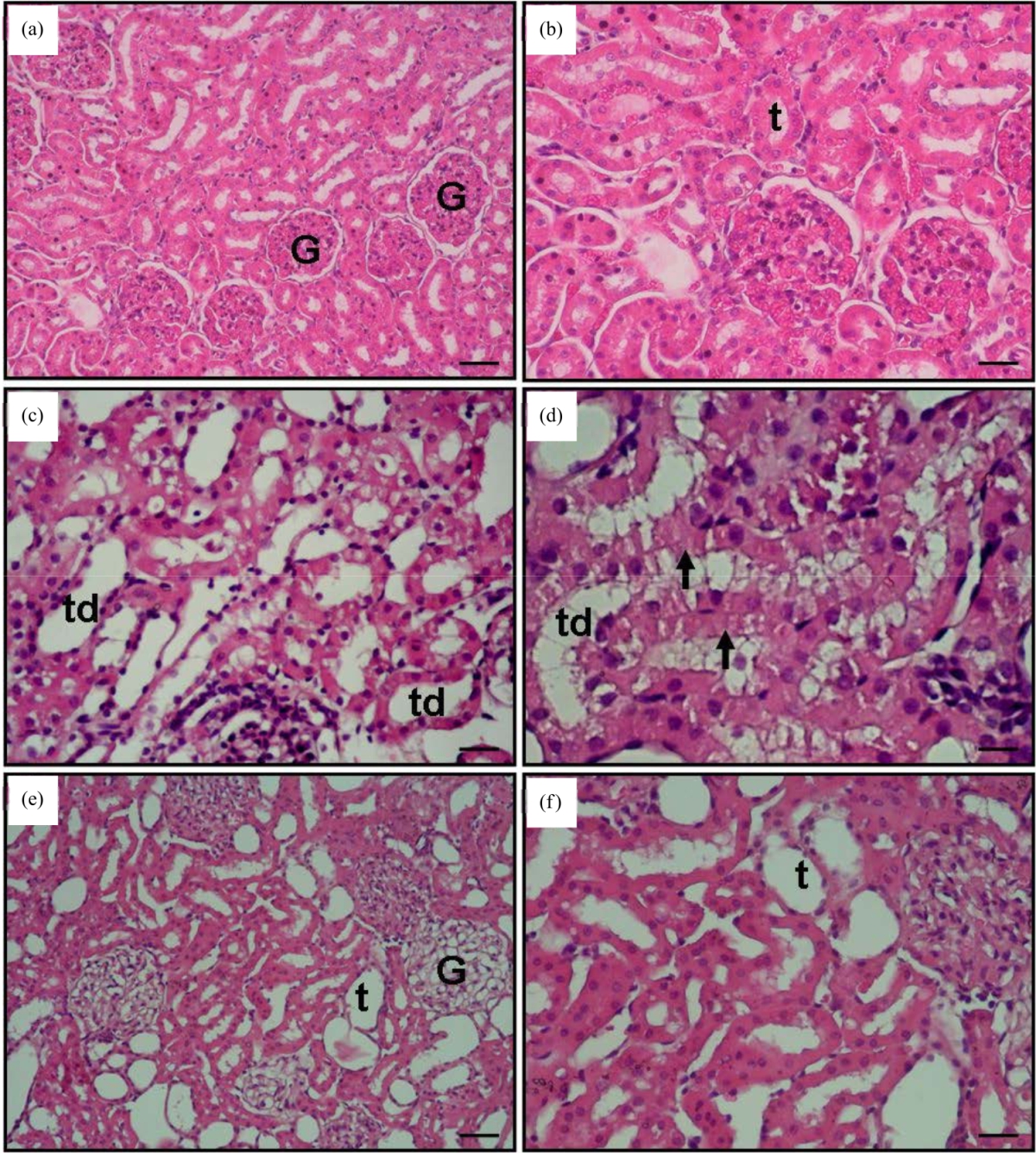
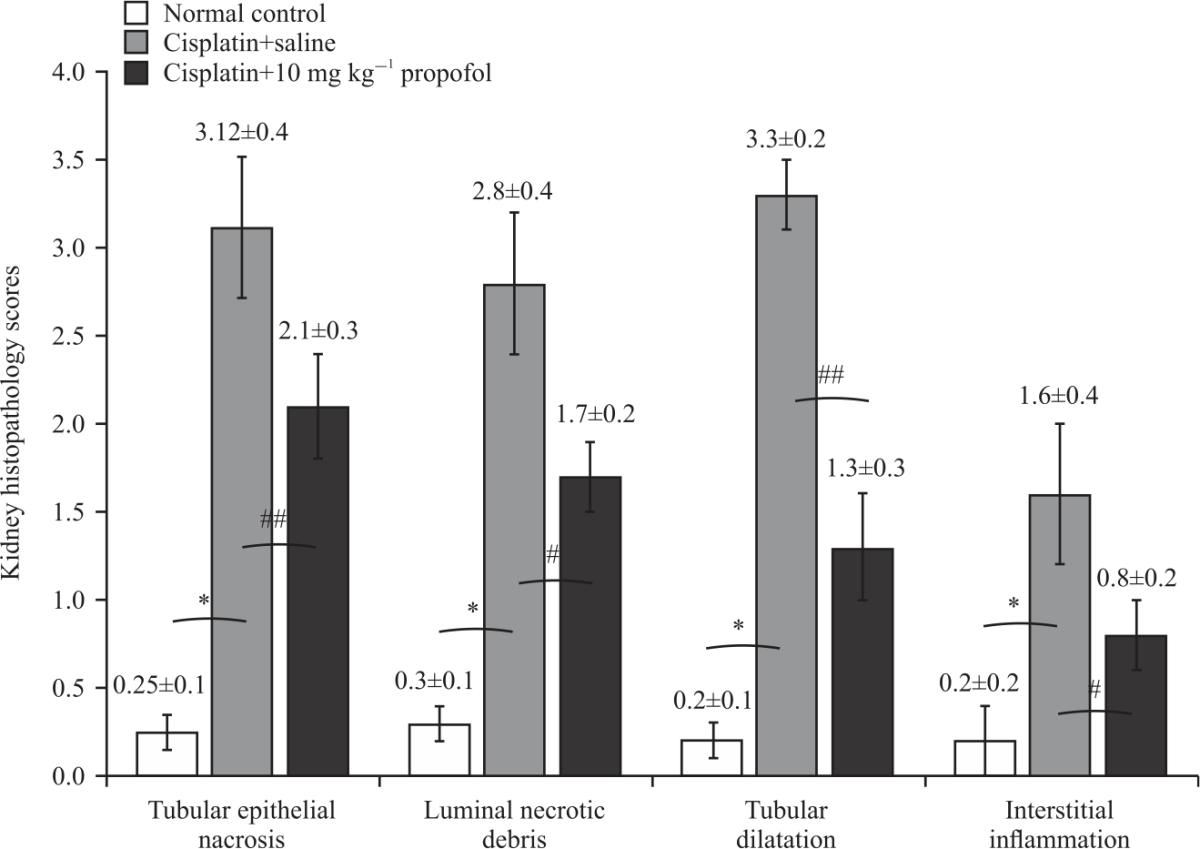
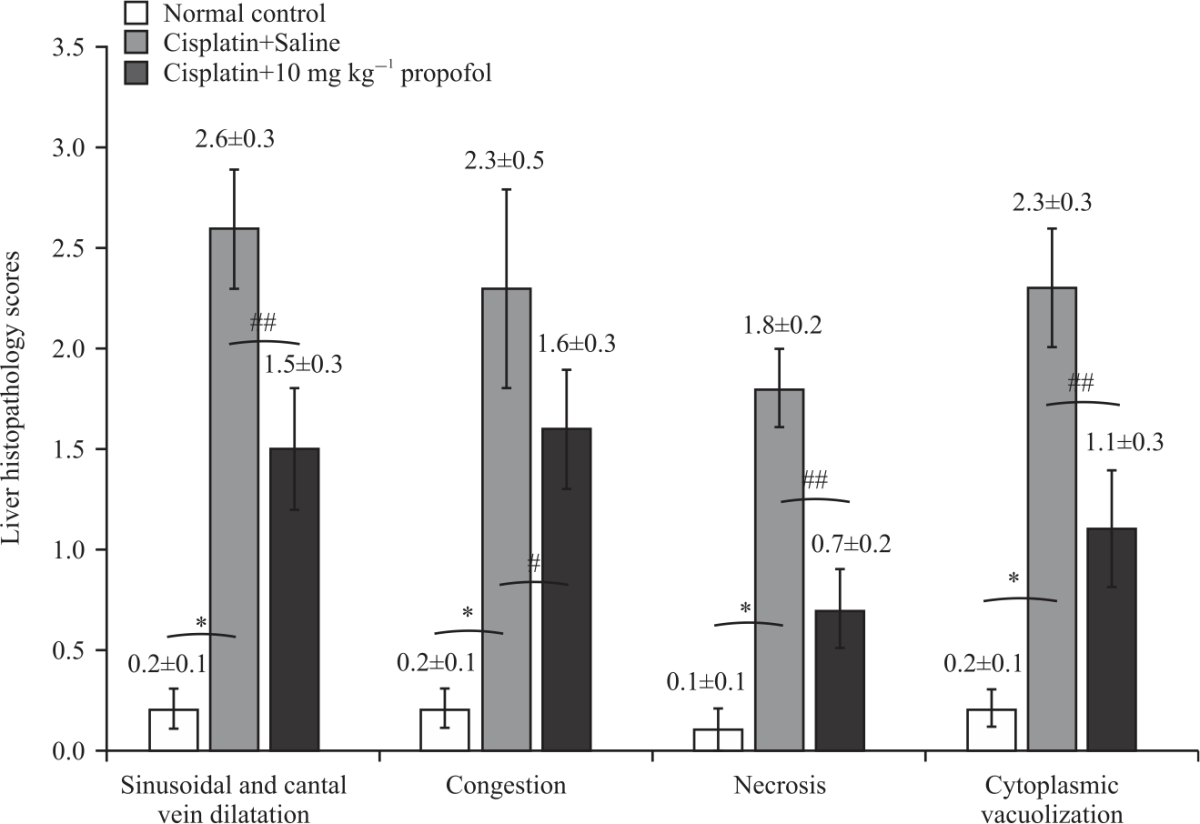
Research Article
Ameliorating Effect of Propofol on Cisplatin-Induced Liver and Kidney Damage in Rats
Bagcılar Research and Training Hospital, Department of Medical Oncology, Merkez Mah, Dr. Sadık Ahmet Cd., Bagcılar, Istanbul, Turkey
Bahattin Ozkul
Department of Radiology, Atlas University, Hamidiye Mah, Anadolu Cd. No: 40, Kagıthane 34408, Istanbul, Turkey
Mumin Alper Erdogan
Department of Physiology, Faculty of Medicine, Izmir Katip Celebi University, Havaalani Sosesi Cd., No: 33/2, Izmir, Turkey
LiveDNA: 90.25645
ORCID: 0000-0003-0048-444X
Oytun Erbas
Department of Physiology, Demiroglu Bilim University, Izzetpasa Mah, Abide-i Hurriyet Cd. No: 166, Sisli 34381, Istanbul, Turkey