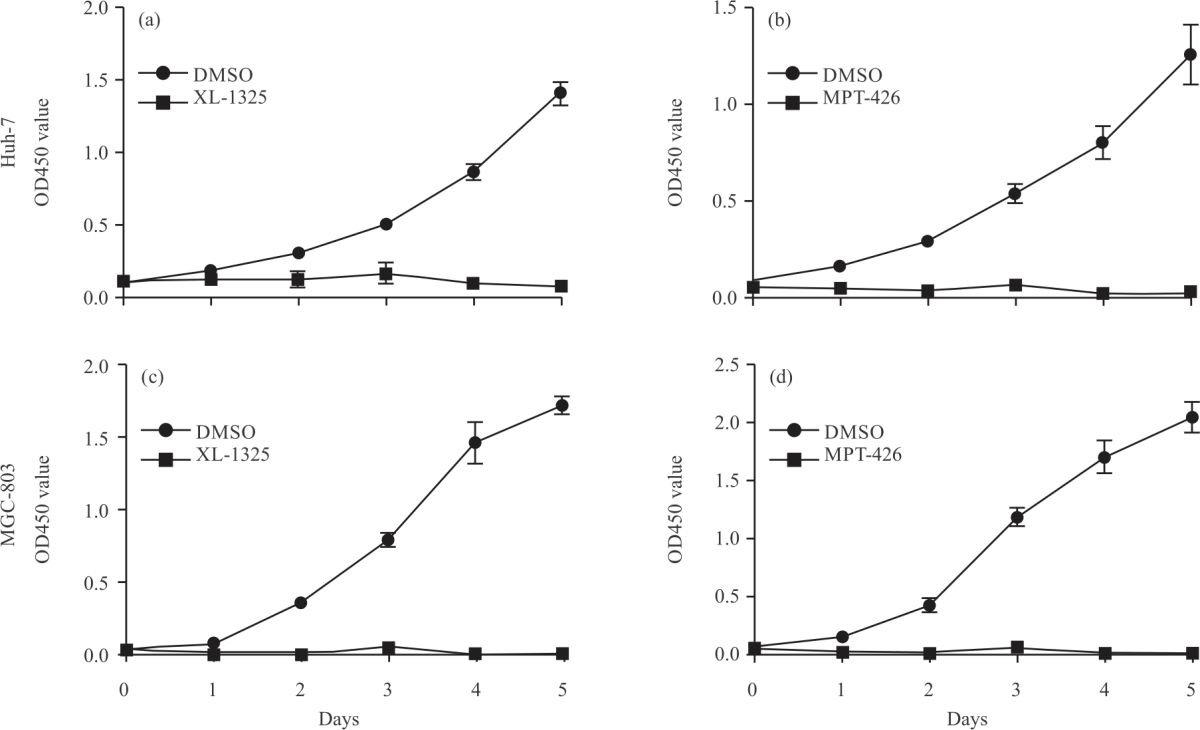
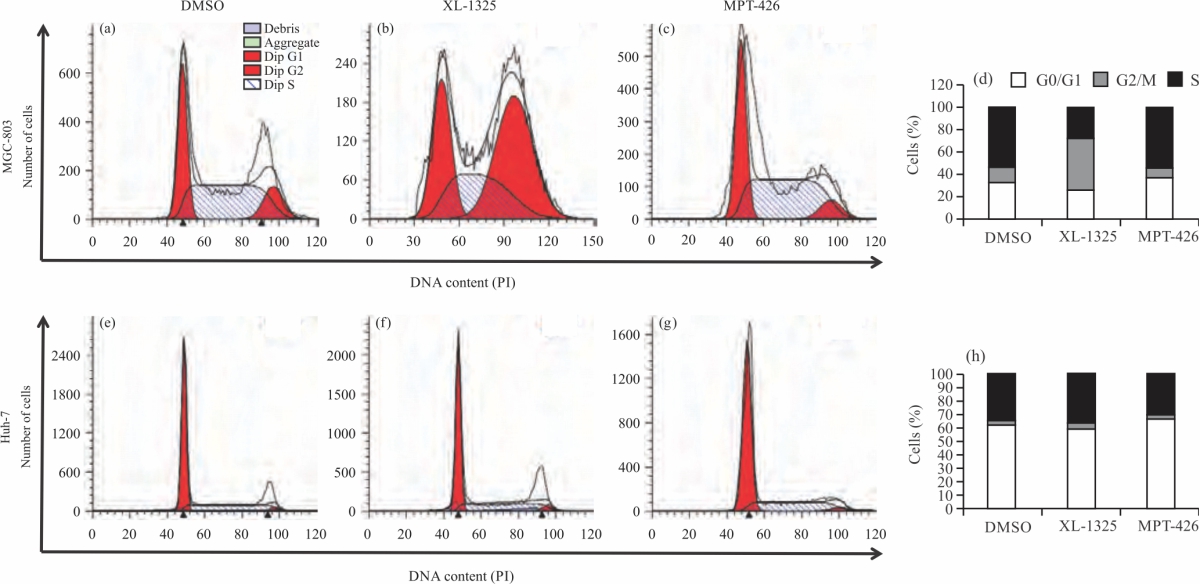
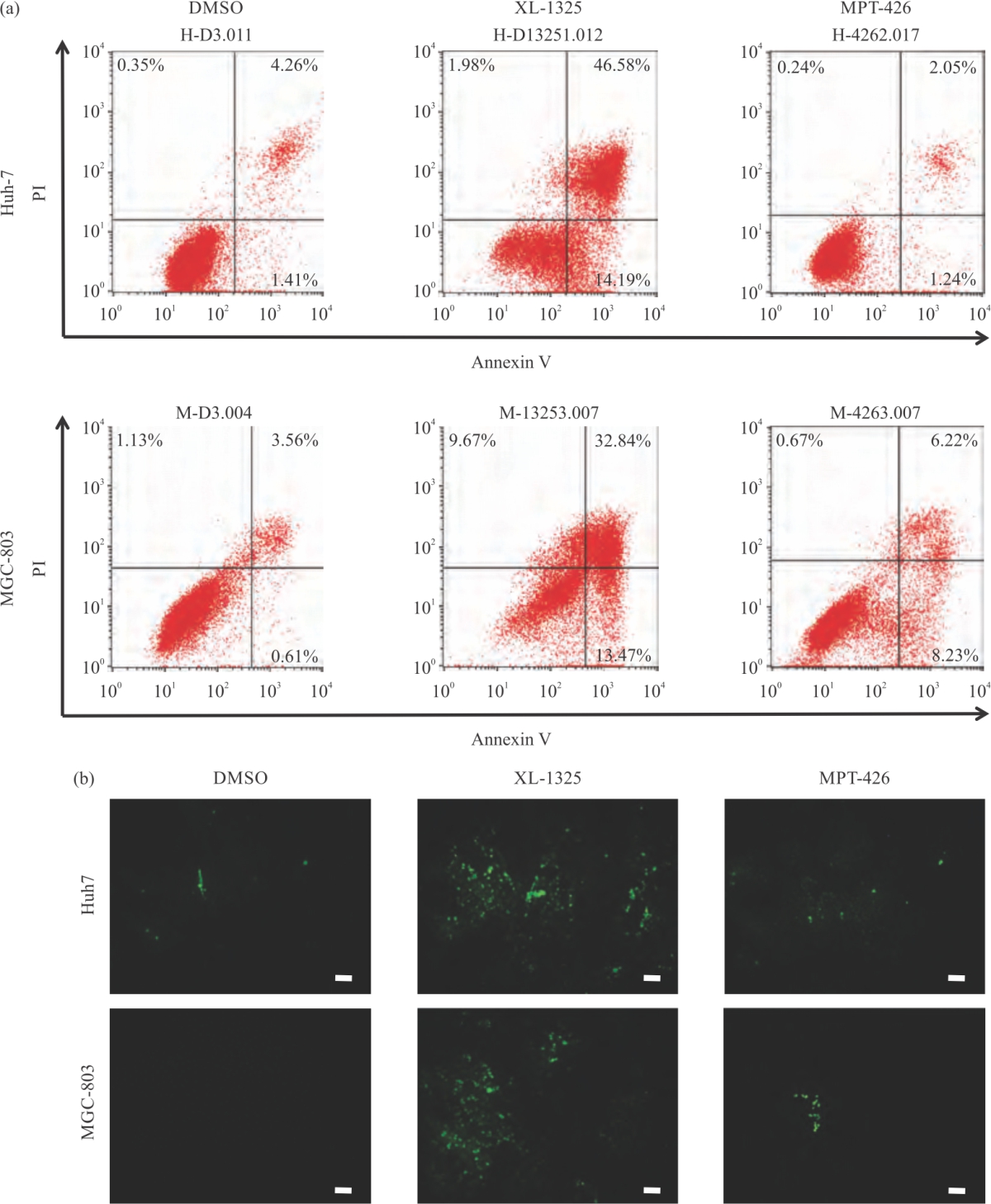
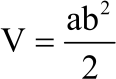Research Article
Acute Anti-Cancer Activity of Crude Extracts from two Endophytic Fungi Chaetomium cochliodes and Penicillium Sp. in Cancer Cell Lines and Mice
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
Qinghua Ma
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
Ruili Cai
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
Jingya Miao
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
Zexuan Yan
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
Xiaolong Yang
School of Pharmaceutical Sciences, South-Central University for Nationalities, Wuhan, People’s Republic of China
Yemiao Chen
Institute of Pathology and Southwest Cancer Center, Southwest Hospital, Army Medical University, Chongqing, People’s Republic of China
LiveDNA: 86.37953
ORCID: 0000-0001-5397-1407