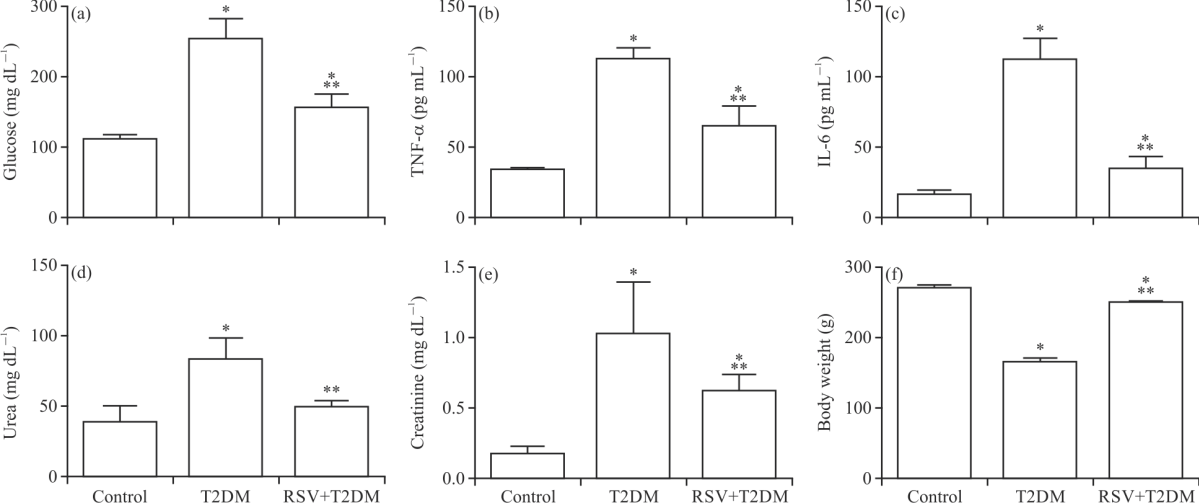
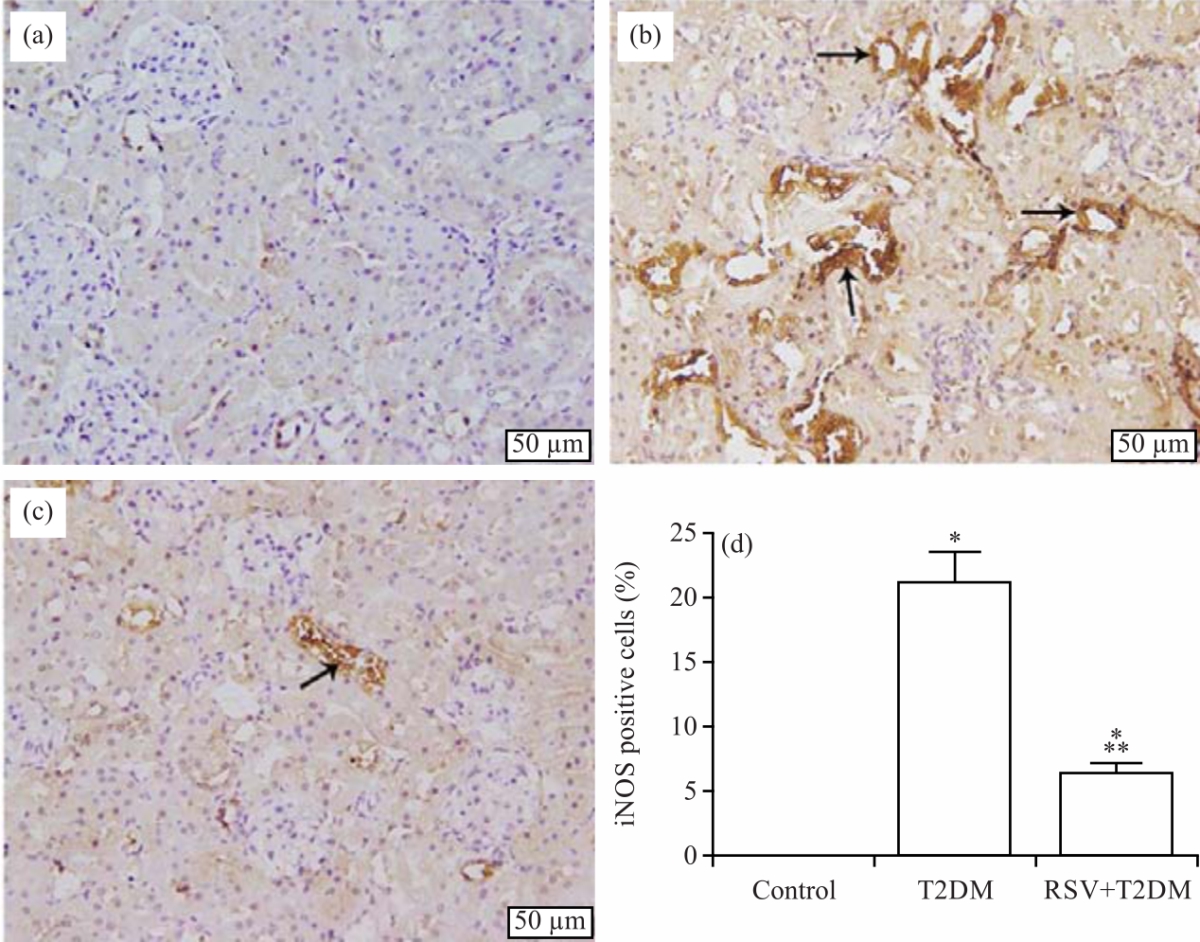
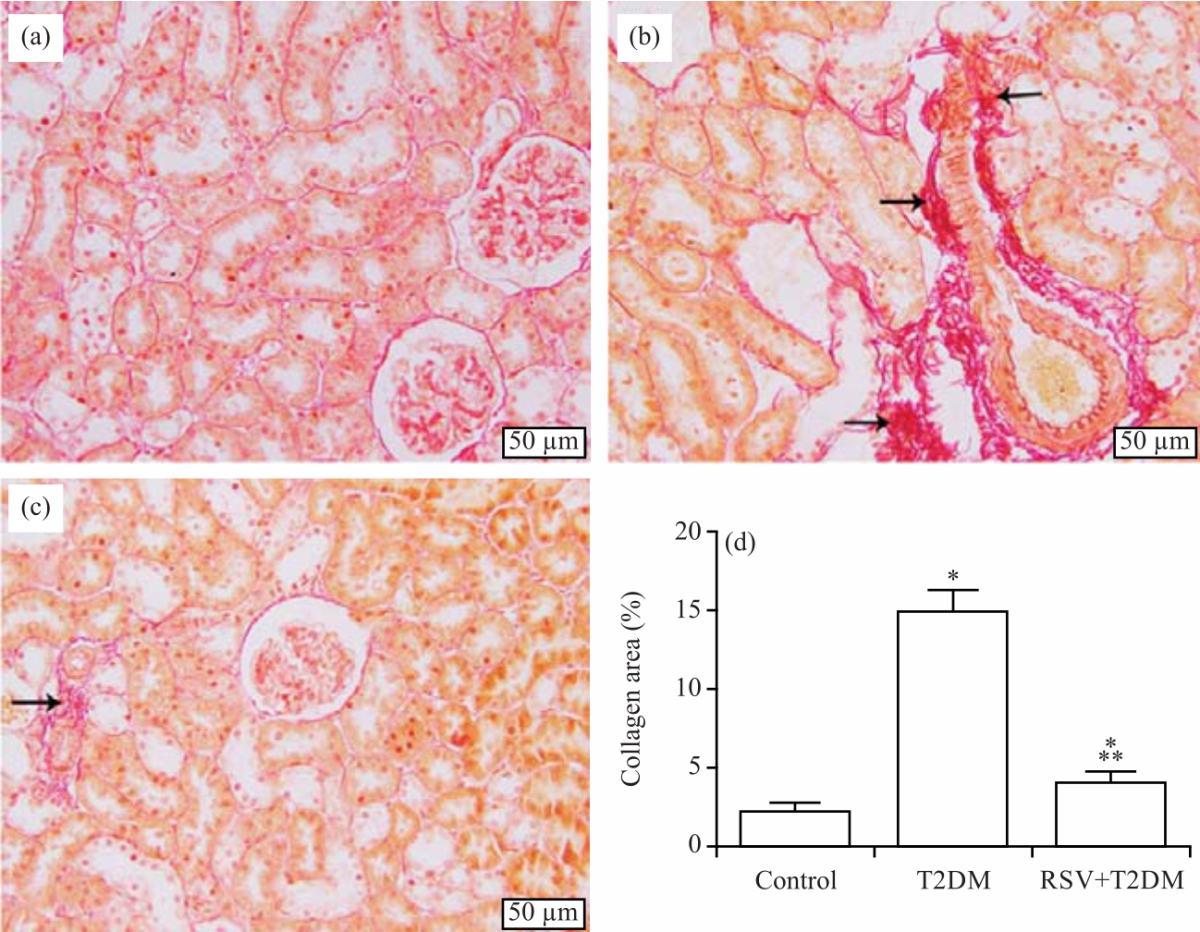
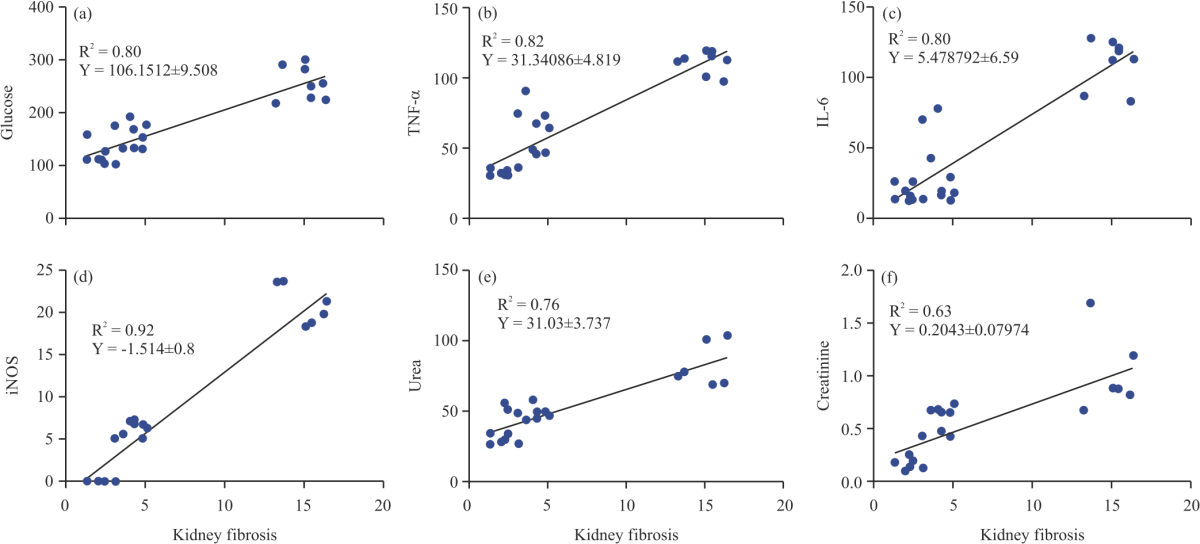
Research Article
Resveratrol Ameliorates Kidney Injury and Fibrosis Secondary to Diabetes in Association with Inflammation and Nitrosative Stress Inhibition in Rats
Department of Biology, College of Science, Princess Nourah bint Abdulrahman University, P.O. Box 84428, Riyadh 11671, Saudi Arabia
LiveDNA: 966.15453
ORCID: 0000-0002-1827-0995