Research Article
Doxorubicin Attenuates BDNF mRNA Expression in Hippocampal Neuronal Cells
Department of Pharmacology and Toxicology, College of Pharmacy, Qassim University, 51452 Al Qassim, Kingdom of Saudi Arabia
LiveDNA: 966.30951
Brain-Derived Neurotrophic Factor (BDNF) belongs to a family of neurotrophins controlling several physiological functions such as survival, differentiation, neurogenesis and synaptogenesis1. BDNF plays an important role in regulating activity-dependent forms of synaptic plasticity in the Central Nervous System (CNS). The physiological responses of BDNF are mediated by activation of the tropomyosin-related kinase B (TrkB) receptor that belongs to the tropomyosin-related kinase (Trk) family2. The role of BDNF in cognitive function, learning and memory formation has been established. Studies in rodent models have shown that BDNF mRNA expression increased in the hippocampus following training in the Morris Water Maze (MWM)3 radial arm maze4 and contextual fear conditioning5. In contrast, decreased BDNF mRNA expression was associated with memory impairment caused by dopamine transporter knockout in mice6. As further proof that BDNF activity regulates hippocampal learning and memory processes, BDNF protein showed the highest expression levels in the hippocampus, cortex, striatum and amygdala7. All these regions of the brain play a role in cognitive function8. For instance, BDNF mRNA and protein levels and TrkB receptor phosphorylation were increased in the amygdala following fear conditioning9. Additionally, intrahippocampal administration of BDNF improved memory function in MWM10, however, administration of anti-BDNF antibodies leads to memory impairment in MWM11. Together, these studies reveal that BDNF plays an essential role in memory consolidation and synaptic plasticity.
Doxorubicin is an anticancer drug that belongs to the anthracycline drug class. It is frequently used to treat different types of cancer such as breast, prostate and lung cancers12. Doxorubicin acts as an anticancer agent via different mechanisms such as DNA intercalation, topoisomerase II inhibition and increased Reactive Oxygen Species (ROS) formation13. Doxorubicin is known as a drug that cannot cross the Blood-Brain Barrier (BBB)14, however, it is widely reported to induce cognitive impairment15,16. The link between doxorubicin and BDNF mRNA expression in the brain has not been elucidated thus far. It is reported that doxorubicin decreases BDNF mRNA expression in the heart17, however, because of BBB selectivity, the concentration of doxorubicin that reaches the heart is higher than that which accesses the brain18,19. Therefore, the effect of doxorubicin on the brain at low concentrations (250, 500 and 1000 nM) was assessed in this study.
This study aimed to evaluate the effect of doxorubicin treatment on the survival of hippocampal neurons and BDNF mRNA expression as a potential mechanism of doxorubicin-induced cognitive impairment.
Study area: The research study was conducted in the College of Pharmacy, Qassim University, Kingdom of Saudi Arabia, from January 5-30, 2021.
Chemicals: Doxorubicin was obtained from Cell Signaling Technology (Danvers, MA, USA) and dissolved in dimethyl sulfoxide (DMSO). Dulbecco’s Modified Eagle’s medium (DMEM), fetal bovine serum, G-418, puromycin, poly-L-lysine and 3-(4, 5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Trypsin-EDTA and Neuronal Protein Extraction Reagent (N-PER) were purchased from Thermo Fisher Scientific Inc. (Rockford, IL, USA). BDNF mRNA was purchased from Life Science, Bio-Rad (Hercules, CA, USA).
H19-7 hippocampal neurons: Rat embryonic hippocampal neuronal (H19-7/IGF-IR) cells were purchased from ATCC (Manassas, VA, USA). They were grown on poly-L-lysine-coated 6-well plates in DMEM adjusted with 10% fetal bovine serum, 200 Lg mL–1 G-418 and 1 Lg mL–1 puromycin at 34°C as described previously20. The neurons were exposed to three different concentrations of doxorubicin for 12 hrs. Cells were spread in 10 cm2 poly-L-lysine-coated tissue culture plates and harvested by trypsinization [trypsin 0.25% (w/v) plus EDTA] after cells reached 80% confluence (2-3 day), then plated into poly-L-lysine-coated 6-well plates at a density of 20,000 cells/well. Cells were incubated at 34°C and supplemented with 5% CO2. The cell cultures were used within 10 passages after the cells were received. The doxorubicin treatment was added when the neuronal cells were over 70% confluent in the plates for 12 hrs before extraction.
MTT reduction assay: The viability of cells was determined by MTT assay. Hippocampal (H19-7) cells were plated on 24-well culture plates at a density of 2000 cells per well. Cells were cultured in the medium for 36 hrs, following which the medium was replaced with either medium only or medium containing doxorubicin for 12 hrs. The medium was then removed, mixed with a solution of 1 mg mL–1 MTT and incubated for 4 hrs in a 5% CO2 humidified atmosphere at 34°C. The supernatant was removed and the formazan crystals in the cells were solubilized with DMSO. Absorbance at 570 nm was determined for each well.
Real-time quantitative PCR analysis: Samples were obtained from hippocampal neuronal extracts of control and treated cells. Total RNA was extracted from tissue samples using the TRIzol reagent (Sigma-Aldrich, St. Louis, MO, USA) and the RNeasy kits (Qiagen, Hilden, Germany). Subsequently, total RNA was treated with RNase-free DNase (Ambion, Carlsbad, CA, USA) to eliminate possible traces of genomic DNA. For the synthesis of first-strand complementary DNA (cDNA), 500 ng of total RNA was reverse transcribed using a cDNA synthesis kit (Applied Biosystems, Foster City, CA, USA). The cDNA samples were then amplified by PCR using 2.5 U of Taq DNA polymerase (Qiagen, Shanghai, China). Real-time PCR was performed on an iCycler iQ5 detection system (Bio-Rad, Hercules, CA, USA) with SYBR Green reagents (Bio tool, Stratech Scientific Ltd., Cambridge shire, UK). The primer sequences are BDNF Forward: CTG GCG GTT CAT AAG GAT AG, BDNF Reverse: GCG TAA GTT CAA GGT GTC A, GAPDH Forward: GTC TAC TGG CGT CTT CAC, Reverse CAC CAC CTT CTT GAT GTCA. Advanced SYBR Green Supermix (Bio-Rad) was used for real-time quantitation. The reaction conditions were: 95°C for 30 sec, followed by 40 cycles of 95°C for 5 sec and 57°C for 30 sec. After the reaction, the Ct value for each sample was subtracted from the value for the internal control gene to determine ΔCt and 2–ΔΔCt values were compared21.
Statistical analysis: All data are from in vitro studies presented as Mean±SEM (n = 4 experiments) after analysis by one-way analysis of variance (ANOVA) followed by Tukey’s test. Statistics were performed using the GraphPad Prism5 software (San Diego, CA, USA). Statistical differences were considered significant at p<0.05.
MTT reduction assay observation: To assess the effect of doxorubicin on hippocampal neurons, the cells were seeded into 24-well plates and incubated with doxorubicin as described in the methodology. As shown in Fig. 1a, 12 hrs of doxorubicin incubation at 250 nM did not alter the survival rate of the cells, whereas 500 and 1000 nM doxorubicin reduced cell survival by 10 and 20%, respectively.
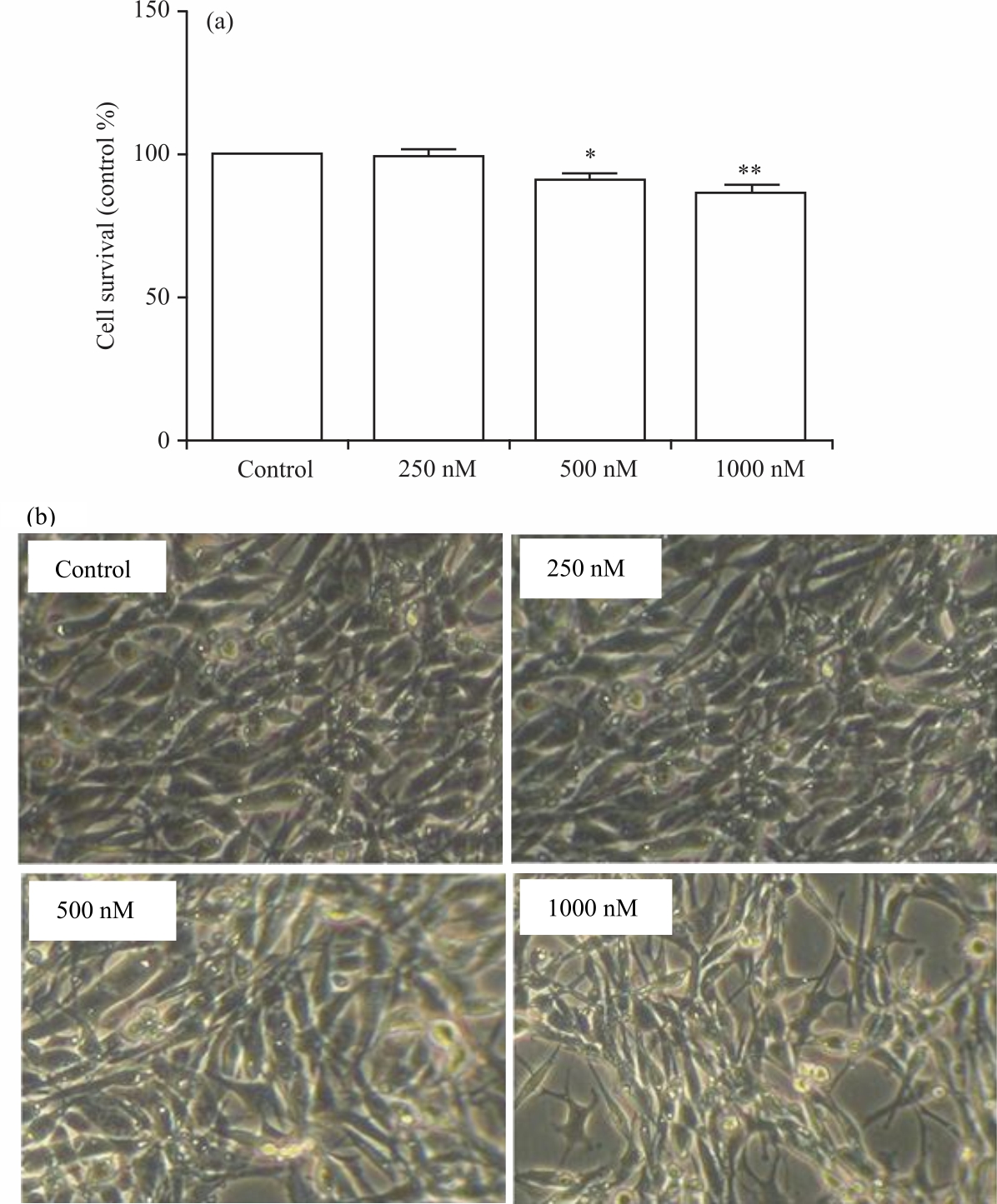 |
| Fig. 1(a-b): | Survival of hippocampal neurons (H19-7) exposed to doxorubicin as shown by MTT assay (a) Changes in survival rate in doxorubicin-treated neuronal and (b) Microscopic image of reduction in the number of cells in the plate |
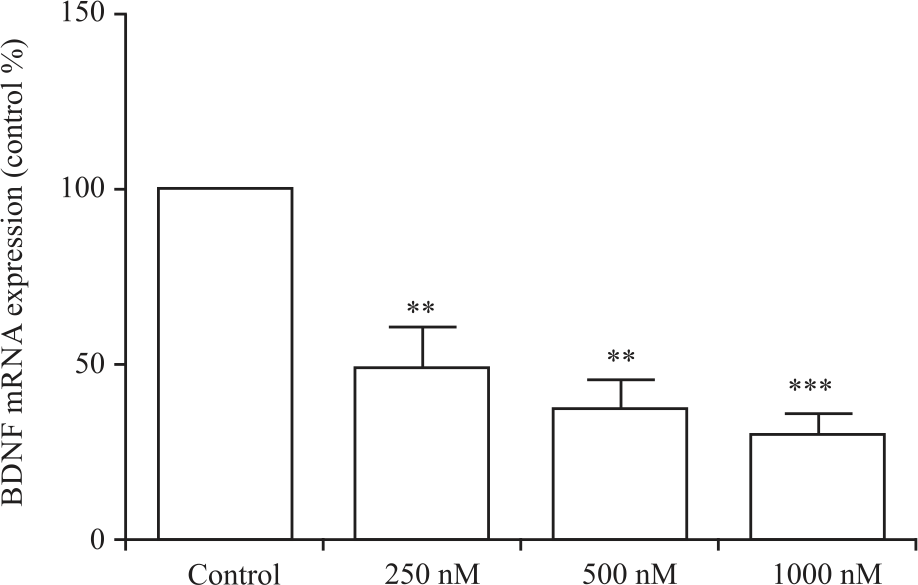 |
| Fig. 2: | Doxorubicin affects the expression of BDNF mRNA in H19-7 cells Cells were treated with doxorubicin for 12 hrs. BDNF mRNA expression analysis by qRT-PCR shows that doxorubicin at 250, 500, and 1000 nM significantly reduces BDNF mRNA expression compared with the untreated control, **p<0.01 and ***p<0.001 |
Figure 1b showed microscopic images of hippocampal neurons after 12 hrs of incubation with doxorubicin. As the concentration of doxorubicin increased in the cells, it caused a decrease in cell division, thus, there were more neurons in the control- and 250 nM-treatments than in the 500- and 1000-nM treatments. BDNF mRNA expression: Next, doxorubicin treatment of H19-7 cells was utilized to determine alterations in BDNF mRNA expression that may explain cognitive impairment following doxorubicin treatment. qRT-PCR analysis revealed that doxorubicin (250, 500 and 1000 nM) decreases the expression of BDNF mRNA in the hippocampal neurons in Fig. 2.
In this study, the effect of doxorubicin exposure on BDNF mRNA expression and cell survival was evaluated using hippocampal neurons. The results revealed that cell survival rates and BDNF mRNA levels were significantly reduced by doxorubicin treatment in a dose-dependent manner. This study was conducted to elucidate the direct effect and molecular mechanism of doxorubicin-induced cognitive impairment. A previous study showed that rat brain slices incubated with doxorubicin showed impaired hippocampal Long-Term Potentiation (LTP)20. LTP is a complex process dependent on several downstream signalling pathways including BDNF22.
Neurogenesis occurs continuously in the hippocampus and dentate gyrus and is well associated with memory formation. Increased neurogenesis is linked with improved behavioural test performance and memory encoding as seen concerning MWM and spatial relational memory23-25. On the contrary, reduced neurogenesis is reported to cause impairment in synaptic plasticity and cognitive functions such as contextual fear conditioning and LTP26. In addition, several studies have reported that chemotherapy reduces neurogenesis in models of chemobrain27,28. The relationship between reduction in BDNF levels and reduction in neurogenesis is well established30-32. Therefore, this study adds to previous work linking chemotherapy, neurogenesis and BDNF by revealing that BDNF mRNA levels were reduced following doxorubicin treatment, which could result in decreased neurogenesis and thus memory impairment.
BDNF also regulates the expression and function of several proteins implicated in synaptic plasticity and cognitive function through regulation of the TrkB receptor32. TrkB is a tyrosine receptor kinase33. When BDNF binds to the extracellular region of the receptor, it increases tyrosine activity, causing autophosphorylation by adenosine triphosphate (ATP)1. Activation of the TrkB receptor activates several other proteins and pathways such as the Mitogen-Activated Protein Kinase (MAPK) and phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) pathways34 as well as regulation of Ca2+/calmodulin-dependent kinase (CaMKII)35. The MAPK and PI3K/AKT pathways and CaMKII protein have been identified as playing an important role in the regulation of cognitive function36,37. Therefore, the reduction in BDNF mRNA levels could result in reduced BDNF protein expression, thereby decreasing the activation of important signalling pathways necessary for memory formation.
The current study aimed to evaluate the neurotoxic effects of doxorubicin on hippocampal neurons. The concentration of doxorubicin used on the neuronal cells was similar to that which may cross the BBB, reach the brain and induce cognitive impairment. The results showed that doxorubicin can reduce cell survival through reduction of BDNF mRNA expression at all three concentrations (250, 500 and 1000 nM) tested in a dose-dependent manner, indicating that this could be a potential mechanism for doxorubicin-induced cognitive impairment.
Studies revealed that the anticancer drug doxorubicin induces cognitive impairment. It was found that doxorubicin treatment potentially induces memory impairment in chemobrain by decreasing brain-derived neurotrophic factor mRNA expression in the hippocampus, in turn decreasing neuronal survival. These findings will assist researchers to overcome chemobrain as a side-effect of cancer chemotherapy.