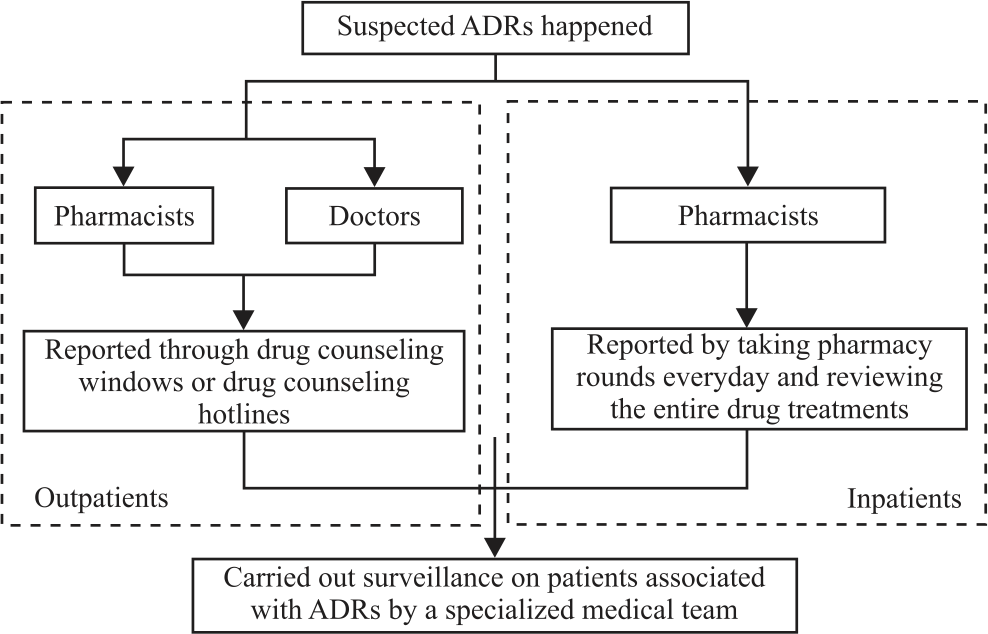
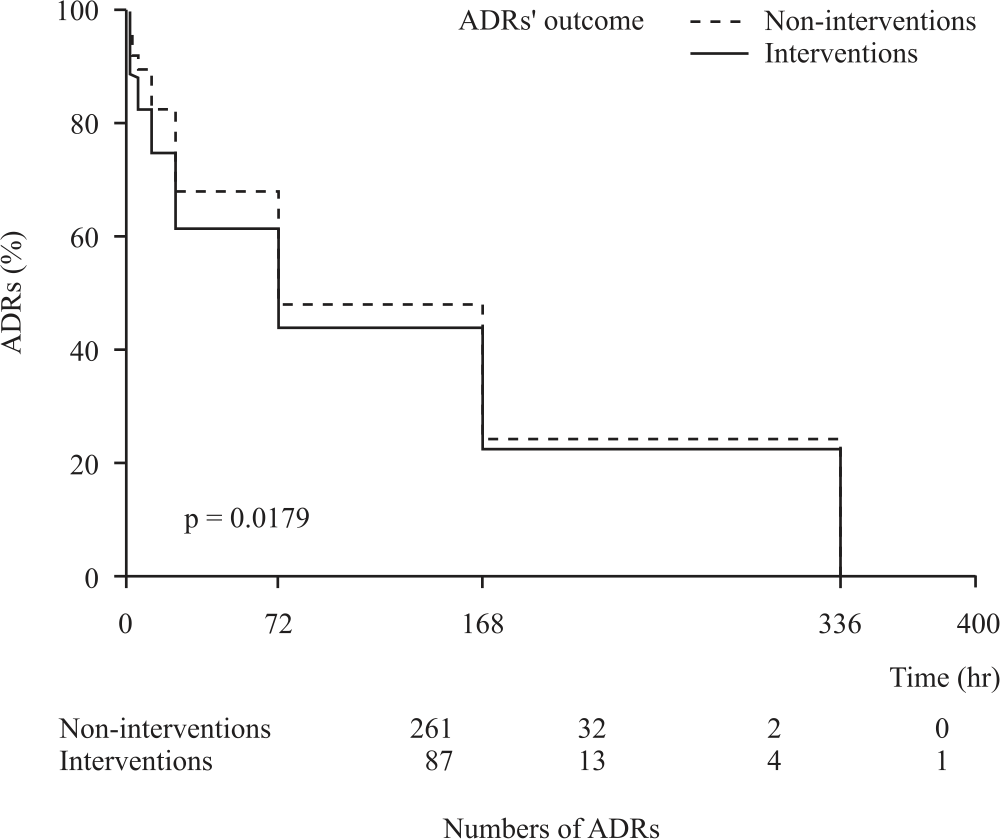
Research Article
Characteristics of Adverse Drug Reactions and Related Risk Factors among Female Patients
Department of Pharmacy, Obstetrics and Gynecology Hospital, Fudan University, Shanghai 200011, China
Yan-Yu Pang
Department of Pharmacy, Obstetrics and Gynecology Hospital, Fudan University, Shanghai 200011, China
Meng-Meng Wang
Department of Pharmacy, Obstetrics and Gynecology Hospital, Fudan University, Shanghai 200011, China
Zhen-Yu Yang
Department of Pharmacy, Obstetrics and Gynecology Hospital, Fudan University, Shanghai 200011, China
LiveDNA: 86.35598