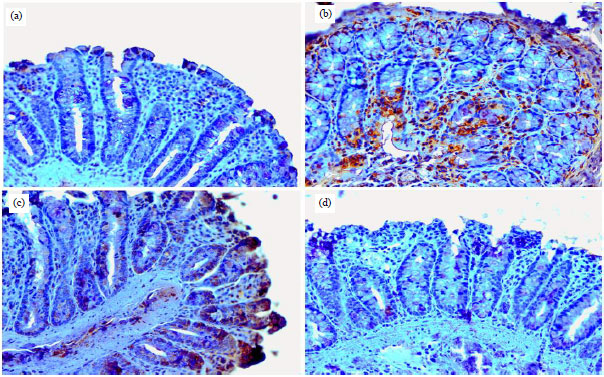
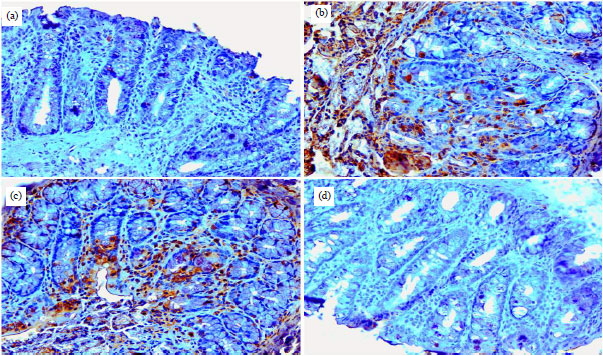
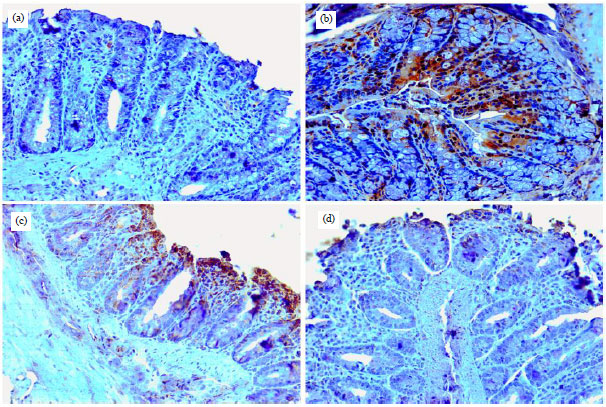
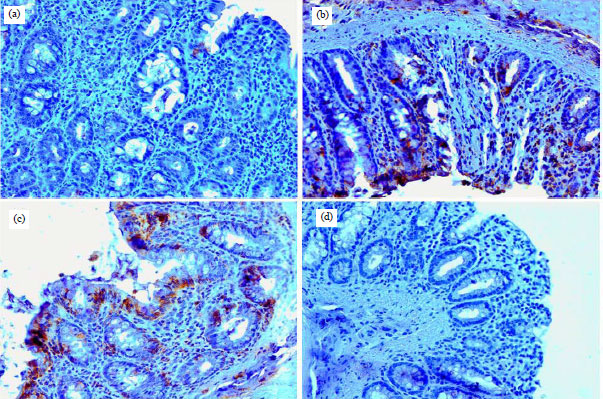
Research Article
Brucine Prevents DMH Induced Colon Carcinogenesis in Wistar Rats
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
Majid Ahmad Ganaie
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
LiveDNA: 91.4319
Basit Latief Jan
Department of Clinical Pharmacy, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia
Hassan Madhkali
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
Mohd Nazam Ansari
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
Najeeb U. Rehman
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
Summya Rashid
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, P.O. Box 173, Al-Kharj, 11942, Saudi Arabia
Muneeb U. Rehman
Department of Clinical Pharmacy, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia