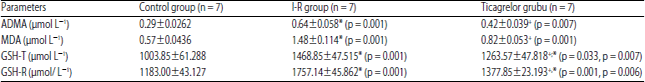Research Article
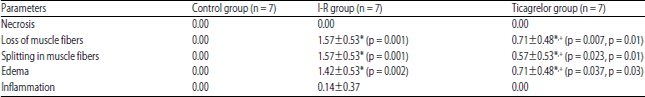
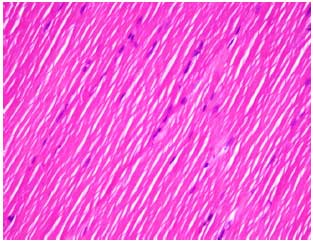
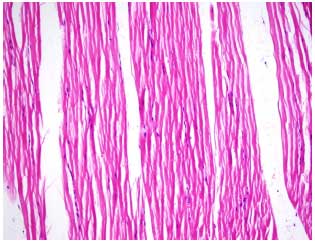
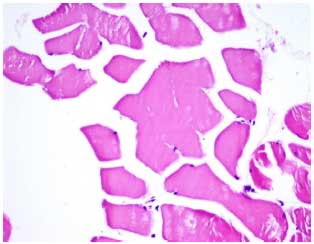
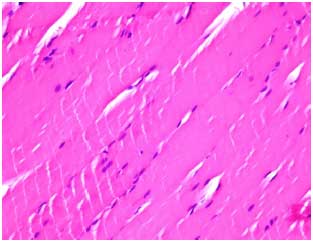
Effects of Ticagrelor on Skeletal Muscle Viability, Asymmetric Dimethylarginine, Malondialdehyde and Glutathione in Ischemia-reperfusion Model
Muş State Hospital, Department of Cardiovascular Surgery, Muş, Turkey
Aydın Kahraman
Siyami Ersek Education and Research Hospital, Department of Cardiovascular Surgery, İstanbul, Turkey
Emre Mutlu
The Ministry of Justice, Council of Forensic Medicine, İstanbul, Turkey
LiveDNA: 90.25281
Emre Elçi
Siyami Ersek Education and Research Hospital, Department of Cardiovascular Surgery, İstanbul, Turkey