Research Article
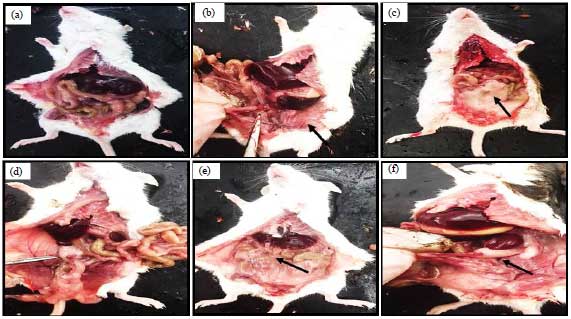
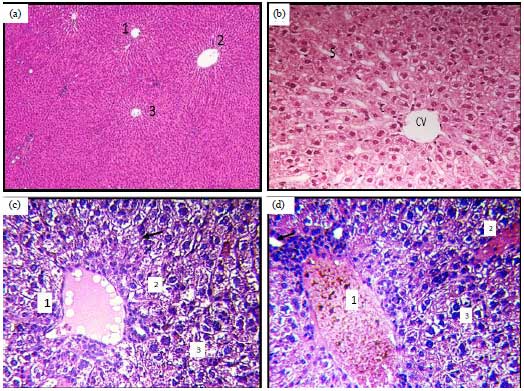
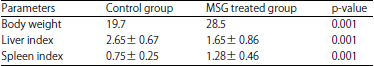
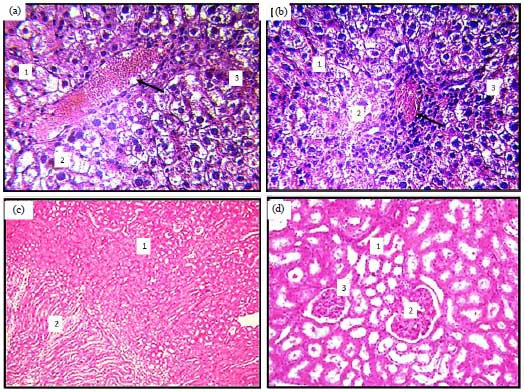
Histomorphological Changes in Mono-sodium Glutamate Induced Hepato-renal Toxicity in Mice
Department of Biology, College of Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
May Bin- Jumah
Department of Biology, College of Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
LiveDNA: 966.25342