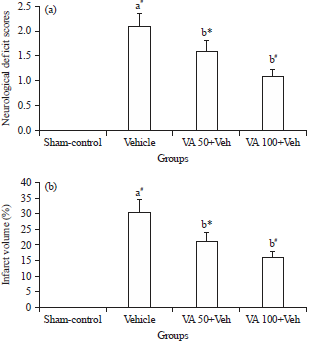
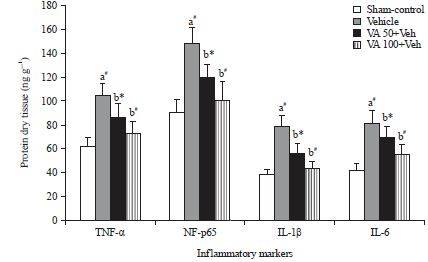
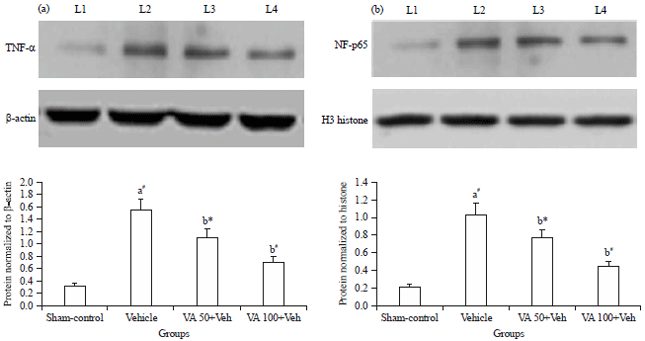
Research Article
Vanillic Acid Improve Neural Function after Focal Cerebral Ischemia-reperfusion Rats
Department of Neurology, Tianjin Nankai Hospital, 300100 Tianjin, China
LiveDNA: 86.5799
Ying Guo
Department of Neurology, Tianjin Nankai Hospital, 300100 Tianjin, China
Shao Yue Zhang
Department of Neurology, Tianjin Nankai Hospital, 300100 Tianjin, China