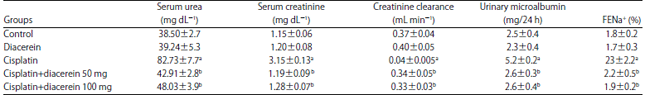
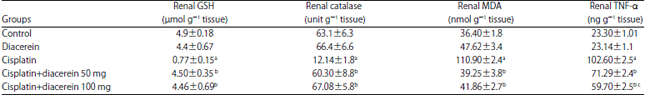
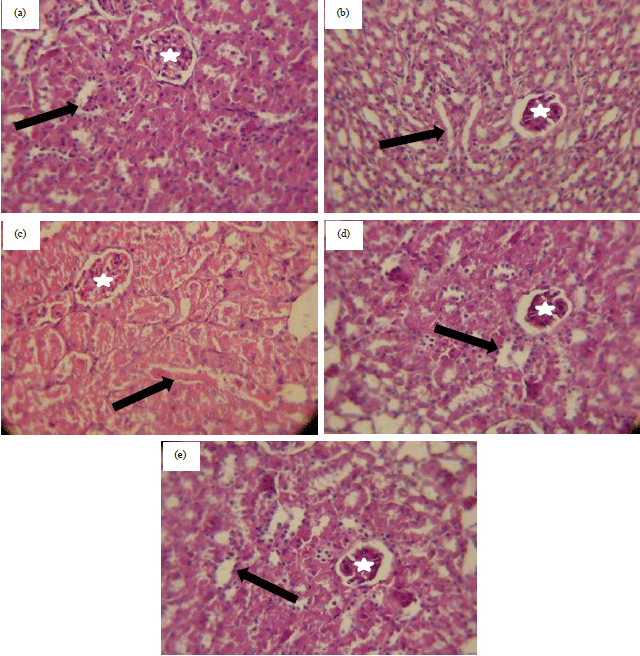
Research Article
Prophylactic Effect of Diacerein against Cisplatin-Induced Nephrotoxicity in Rats
Department of Pharmacology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt
Mohamed Abdellah Ibrahim
Department of Pharmacology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt
Azza Ali El-Shiekh
Department of Pharmacology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt
Nisreen AbdelTawab Osman
Department of Pathology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt
Ayman Geddawy
Department of Pharmacology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt
LiveDNA: 20.19397
Aly Abdelrahman
Department of Pharmacology, Faculty of Medicine, Minia University, 61111 Al-Minia, Egypt