Research Article
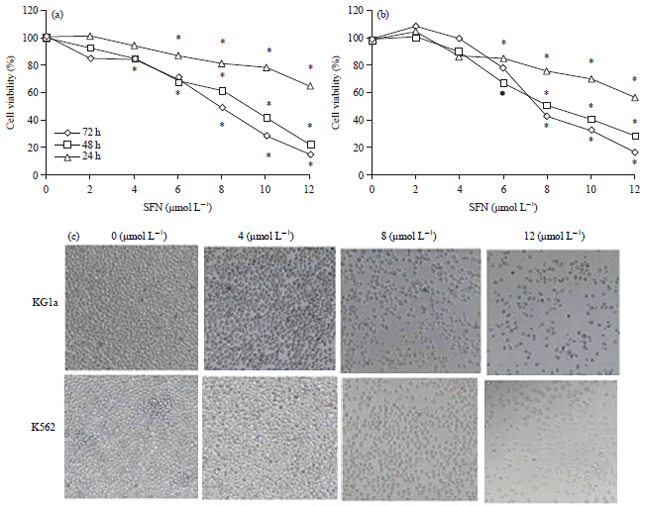
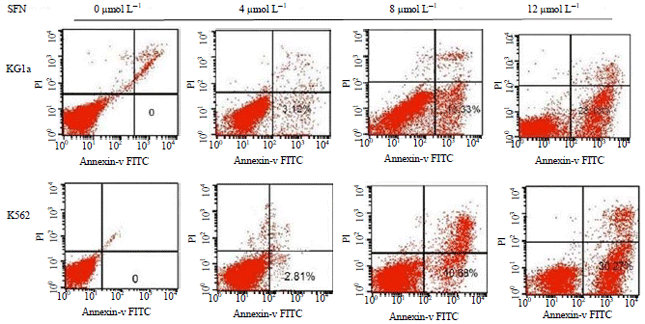
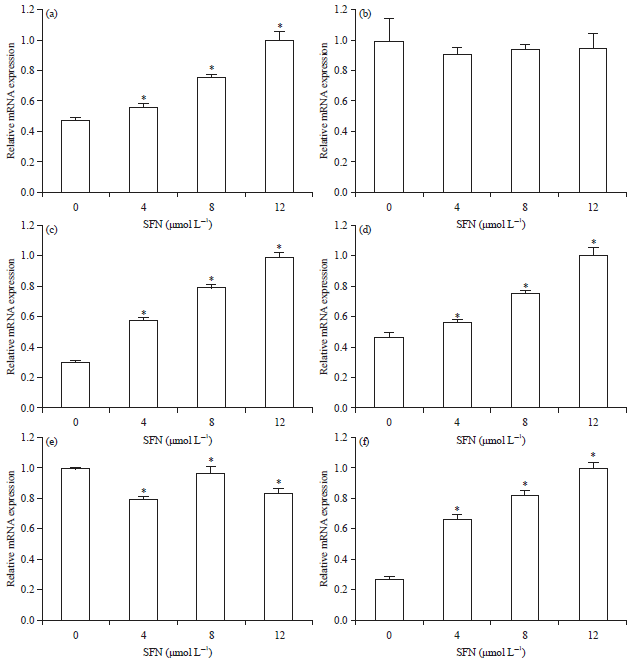
Sulforaphane Induces Apoptosis of Acute Human Leukemia Cells Through Modulation of Bax, Bcl-2 and Caspase-3
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Lulu Chen
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Shanshan Zhu
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Shanshan Wang
Third Affiliated Hospital of Xinxiang Medical University, Xinxiang, 453003 Henan, China
Chen Chen
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Weibin Zhang
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Xia Wang
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Jingjing Zhang
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
Mingyong Wang
School of Laboratory Medicine, Xinxiang Medical University, 453003 Xinxiang, China
LiveDNA: 86.20838