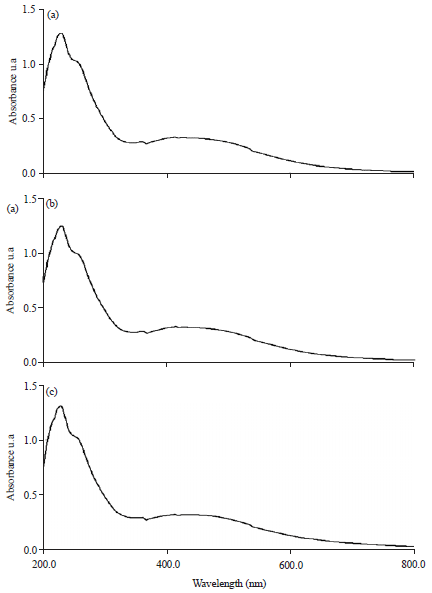
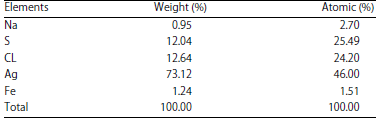
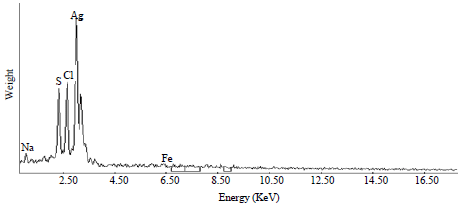
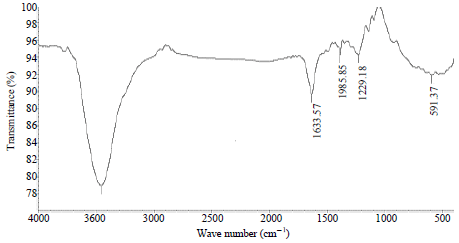
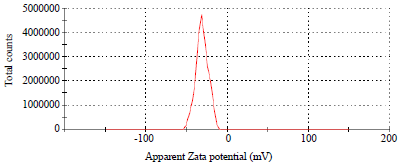
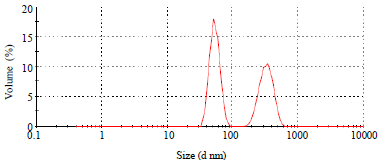
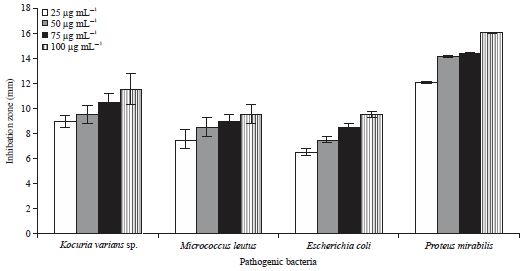
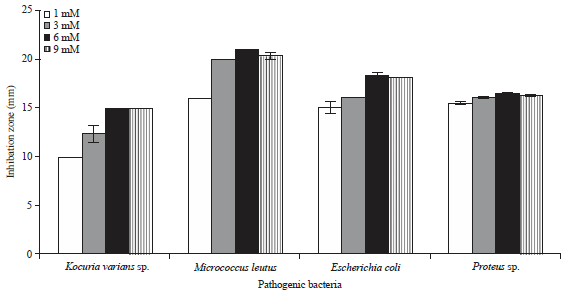
Research Article
Antibacterial Activity of Silver Nanoparticles Using Ulva fasciata Extracts as Reducing Agent and Sodium Dodecyl Sulfate as Stabilizer
Department of Microbial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.16832
Mahmoud Abd El-Mongy
Department of Microbial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.16761
Kamel Fatehy Eid
Department of Microbial Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt
LiveDNA: 20.16831