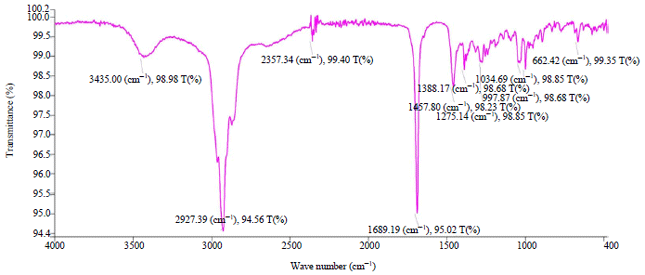
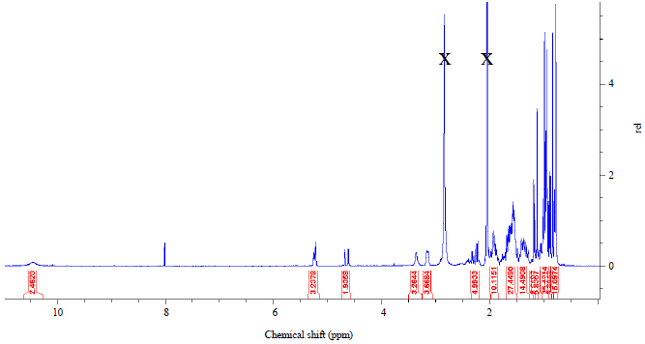
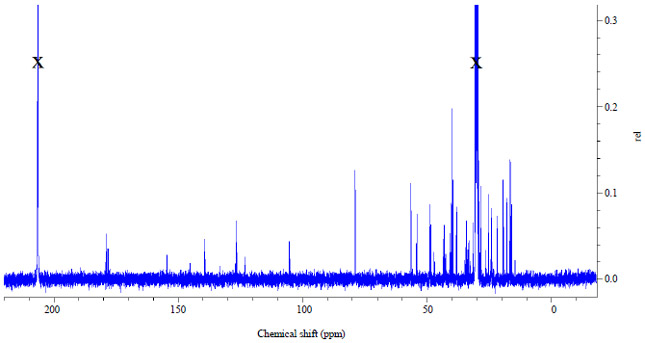
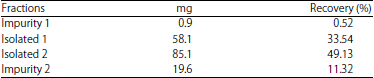Research Article
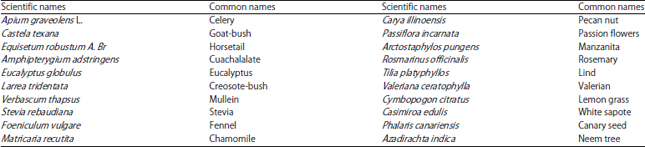
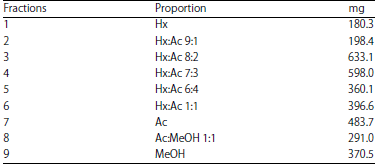
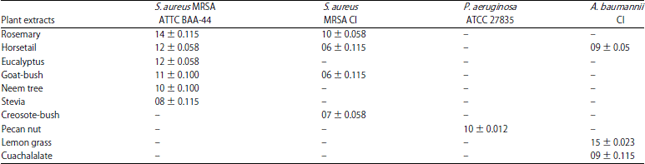
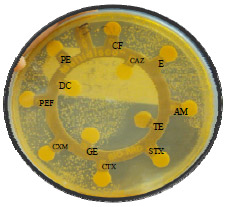
Bactericidal Activity, Isolation and Identification of Most Active Compound from 20 Plants used in Traditional Mexican Medicine Against Multidrug-Resistant Bacteria
Universidad Autonoma de Nuevo Leon, Facultad de Ciencias Biologicas, Laboratorio de Quimica Analitica, Mexico
V.M. Rivas-Galindo
Universidad Autonoma de Nuevo Leon, Facultad de Medicina, Departamento de Quimica Analitica, Mexico
J. Rodriguez-Rodriguez
Tecnologico de Monterrey, Escuela de Medicina y Ciencias de la Salud
S.A. Galindo-Rodriguez
Universidad Autonoma de Nuevo Leon, Facultad de Ciencias Biologicas, Laboratorio de Quimica Analitica, Mexico
C. Leos-Rivas
Universidad Autonoma de Nuevo Leon, Facultad de Ciencias Biologicas, Laboratorio de Quimica Analitica, Mexico
D.G. Garcia-Hernandez
Universidad Autonoma de Nuevo Leon, Facultad de Ciencias Biologicas, Laboratorio de Quimica Analitica, Mexico
LiveDNA: 52.16966