Short Communication
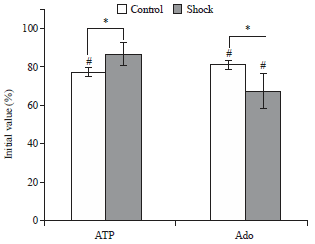
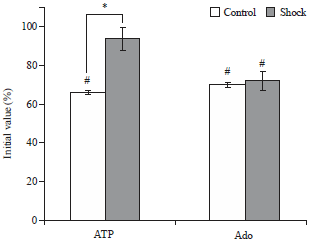
Foot-shock Stimulation Decreases the Inhibitory Action of ATP on Contractility and End-plate Current of Frog Sartorius Muscle
Kazan State Medical University, Kazan, Russia
LiveDNA: 712.6715
Rafis Rustemovich Kamaliev
Kazan State Medical University, Kazan, Russia
Azat Iskhakovich Gabdrakhmanov
Department of Fundamental and Clinical Pharmacology, Kazan Federal (Volga Region) University, Kazan, Russia
Adel Evgenyevich Khairullin
Department of Biochemistry, Kazan State Medical University, Kazan, Russia
Sergey Nikolaevich Grishin
Department of Medical and Biological Physics, Kazan State Medical University, Kazan, Russia