Research Article
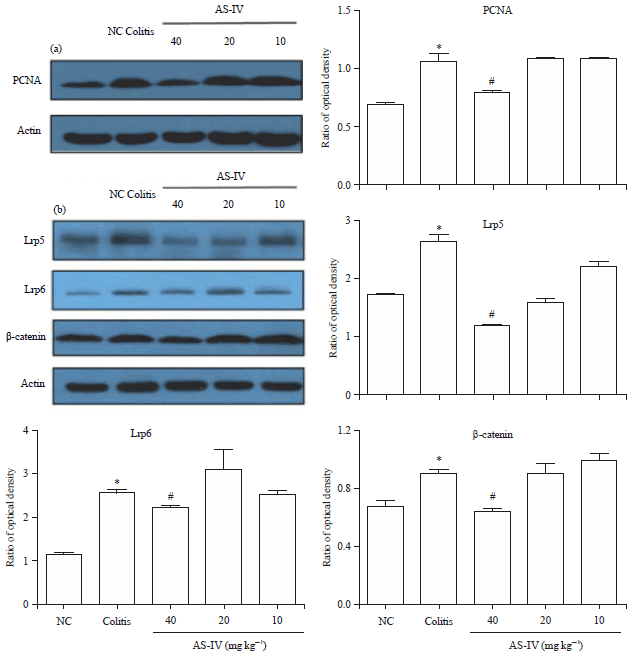
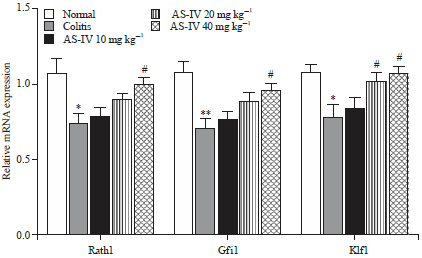
Astragaloside IV Attenuates Trinitrobenzene Sulphonic Acid (TNBS)-Induced Colitis via Improving Mucosal Barrier Function: Role of Goblet Cells
College of pharmacy, Gansu University of Traditional Chinese Medicine, 730000 Lanzhou, China
LiveDNA: 86.24351
Hong-yan Qin
College of pharmacy, Gansu University of Traditional Chinese Medicine, 730000 Lanzhou, China
Hai-Jing Duan
College of pharmacy, Gansu University of Traditional Chinese Medicine, 730000 Lanzhou, China
Qing-lin Ma
Department of Pharmacy, First Hospital of Lanzhou University, 730000 Lanzhou, China
Yuan Ren
College of pharmacy, Gansu University of Traditional Chinese Medicine, 730000 Lanzhou, China
LiveDNA: 86.24378