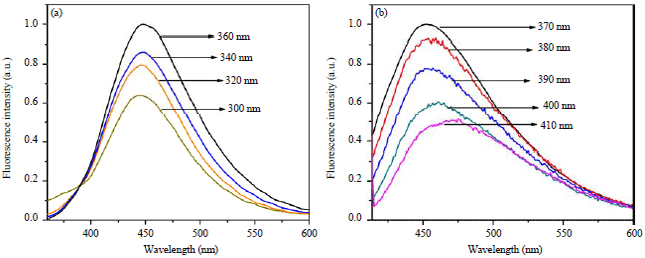
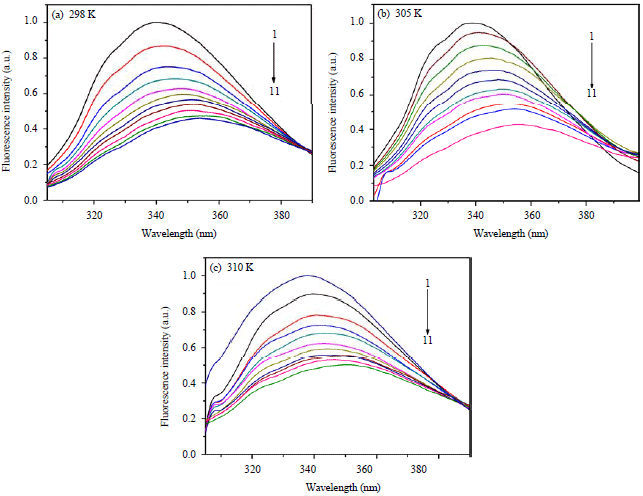
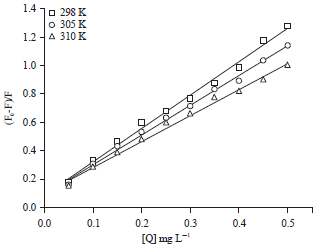
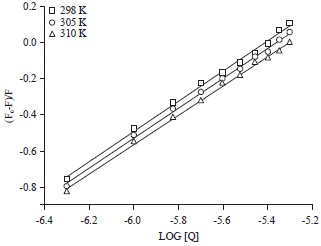
Research Article
Spectral Properties of the Interaction Between Hesperidin of Tangerine Peel’s Active Ingredient with Protein
School of Electrical and Information Engineering, Jiangsu University of Technology, Changzhou City, Jiangsu Province 213001, People�s Republic of China
LiveDNA: 86.21652
Yuxia Sun
School of Electrical and Information Engineering, Jiangsu University of Technology, Changzhou City, Jiangsu Province 213001, People�s Republic of China
Tianyu Chen
School of Electrical and Information Engineering, Jiangsu University of Technology, Changzhou City, Jiangsu Province 213001, People�s Republic of China
Yue Hu
School of Electrical and Information Engineering, Jiangsu University of Technology, Changzhou City, Jiangsu Province 213001, People�s Republic of China