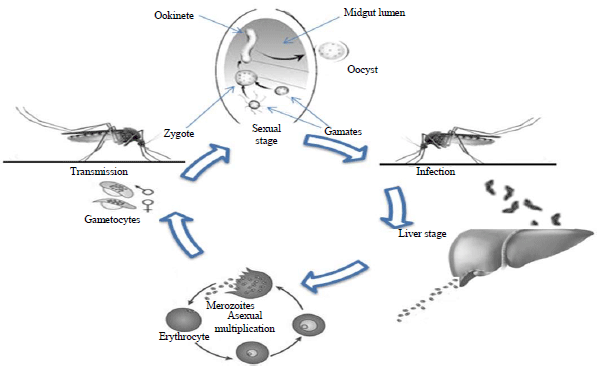
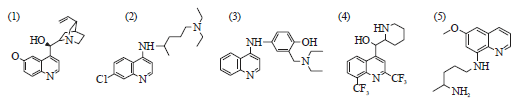
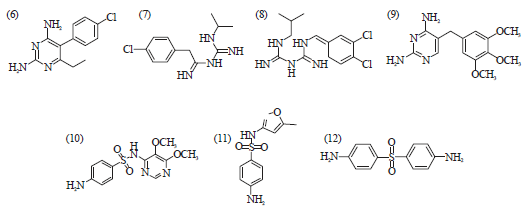
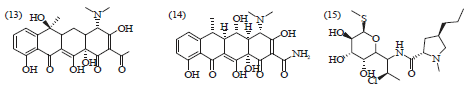
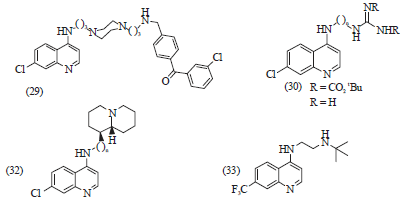
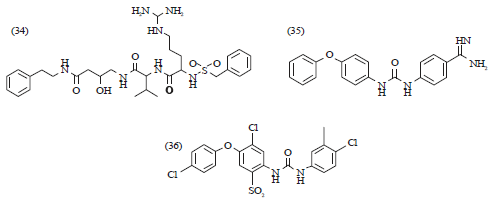
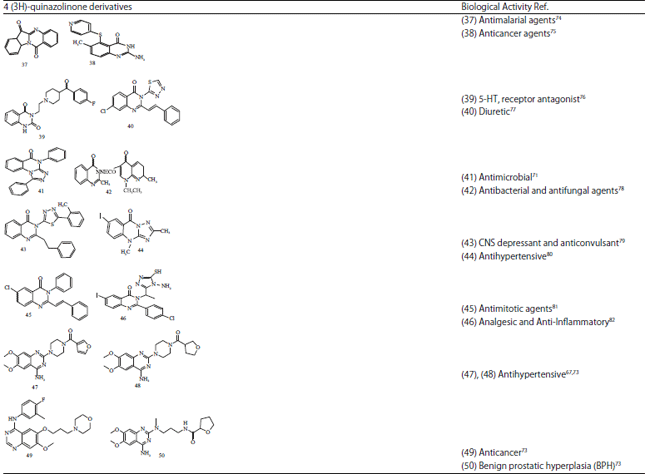
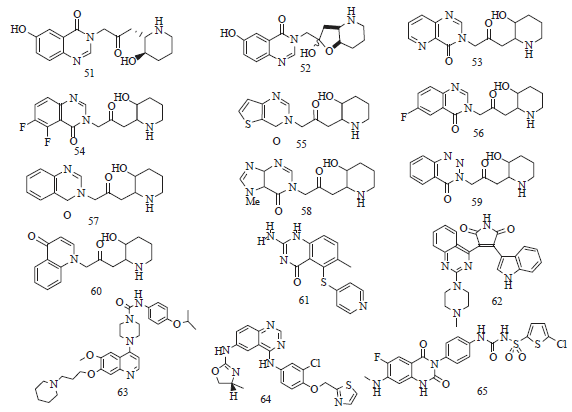
Review Article
Quinazolinone Derivatives as a Potential Class of Compounds in Malaria Drug Discovery
Department of Pharmacy, College of Medicine and Health Sciences, Ambo University, Ambo, Ethiopia
Ishtiaq Ahmed
Department of Biochemistry, University of Agriculture Faisalabad, Pakistan
Faheem Maqbool
School of Pharmacy, Pharmacy Australia Centre of Excellence, University of Queensland, Brisbane, QLD 4102, Australia
Muhammad Anjum Zia
Department of Pharmacy, College of Medicine and Health Sciences, Ambo University, Ambo, Ethiopia