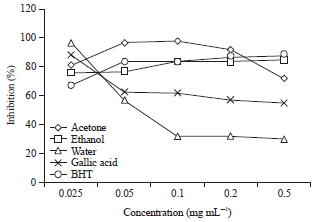
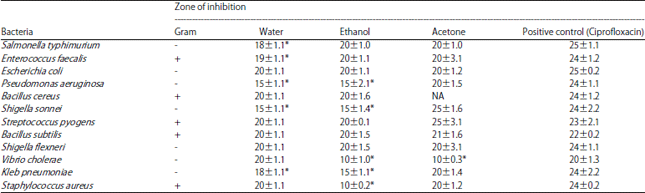
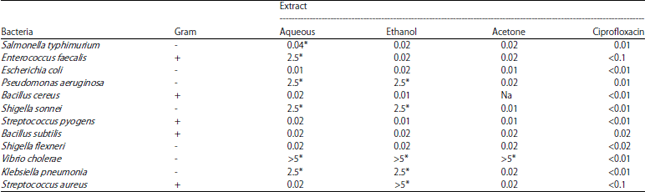
Research Article
Aspects of Medicinal Activities of the Stem Bark Extracts of Curtisia dentata (Burm. F.)
Medicinal Plants and Economic Development (MPED) Research Center, Department of Botany, University of Fort Hare, Alice, South Africa
Anthony Jide Afolayan
Medicinal Plants and Economic Development (MPED) Research Center, Department of Botany, University of Fort Hare, Alice, South Africa