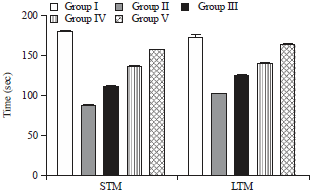
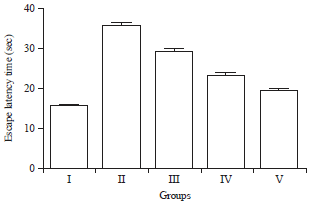
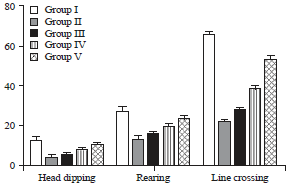
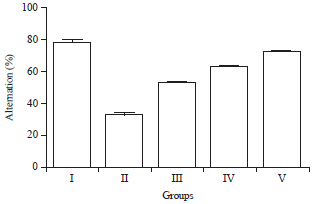
Research Article
Memory and Brain Neurotransmitter Restoring Potential of Hydroalcoholic Extract of Ipomoea aquatica Forsk on Amyloid Beta Aβ (25-35) Induced Cognitive Deficits in Alzheimer’s Mice
Centre for Laboratory Animal Technology and Research, Sathyabama University, Jeppiaar Nagar, Rajiv Gandhi Salai, Chennai, No. 51, Selva Vinayagar Koil Street, Perambur, 600119, Tamil Nadu, India
LiveDNA: 91.1377
P. Panneerselvam
Department of Pharmaceutical Chemistry,
P. Muralidharan
Department of Pharmacology and Toxicology, C.L. Baid Metha College of Pharmacy, Jyothi Nagar, Rajiv Gandhi Salai, Thoraipakkam, Chennai, 600097, Tamil Nadu, India