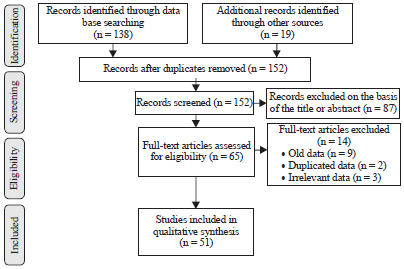
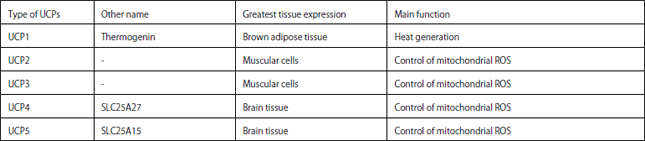
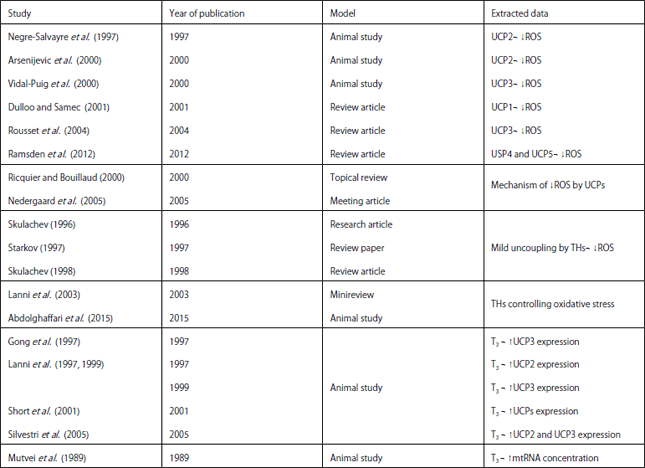
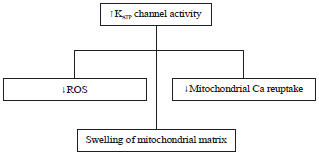
Review Article
Inhibitory Effects of Thyroid Hormones on Mitochondrial Oxidative Stress: A Systematic Review
Department of Toxicology and Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, 1417614411, Iran
Amir Shadboorestan
Department of Toxicology and Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, 1417614411, Iran
Mohammad Abdollahi
Department of Toxicology and Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, 1417614411, Iran
LiveDNA: 98.1833