Research Article
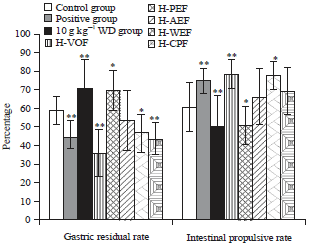
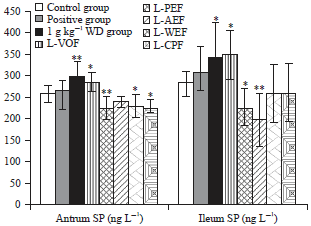
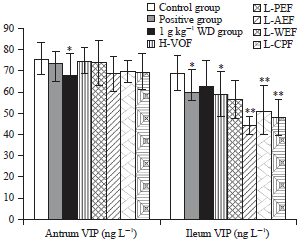
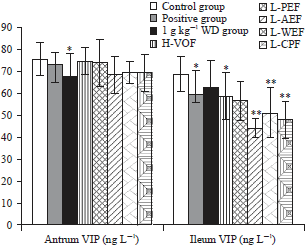
Bidirectional Effective Components of Atractylodis Macrocephalae Rhizoma on Gastrointestinal Peristalsis
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, Dalian 116600, China
XiaLiu
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, Dalian 116600, China
Deqiang Dou
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, Dalian 116600, China