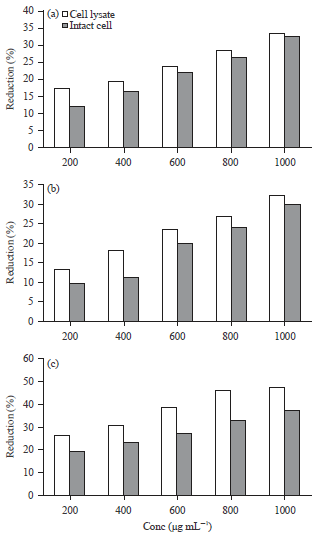
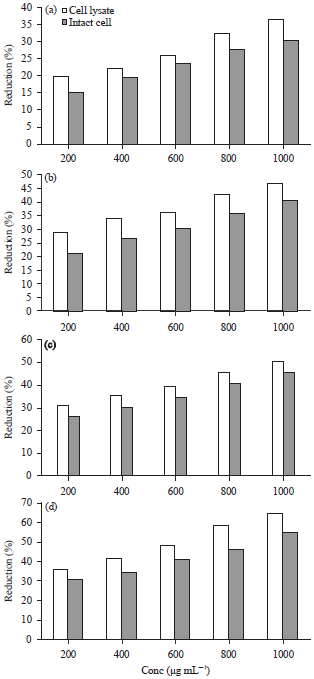
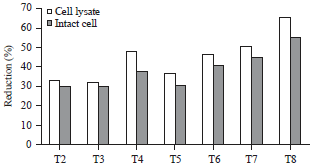
Research Article
In vitro Treatment with Intact Cells or Cell Lysates of Lactobacillus and Spirulina Induced Lowering Effects on Induced Hypercholesteremia
Geenetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Sadat City, Egypt
Ragaa A. Hamouda
Geenetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Sadat City, Egypt
Hoda Mahrous
Geenetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Sadat City, Egypt
Mohammed Labib Salem
Department of Zoology, Faculty of Science, Division of Immunology and Biotechnology, Tanta University, Egypt
Hanafy A. Hamza
Geenetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Sadat City, Egypt
Ekbal Abd Elhafez
National Organization of Drug Control and Research, Egypt