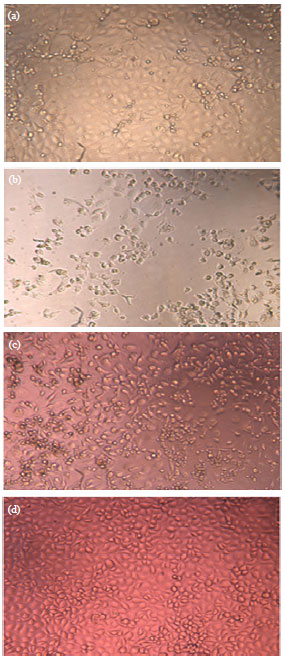
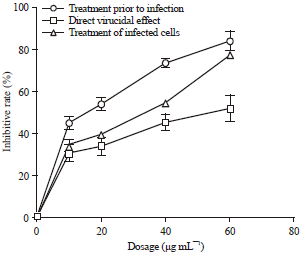
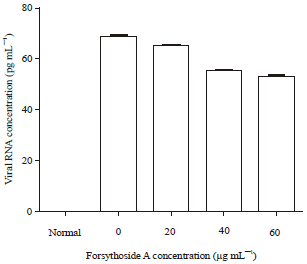
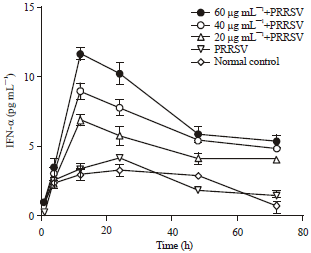
Short Communication
Inhibition of Porcine Reproductive and Respiratory Syndrome Virus in vitro By Forsythoside A
Beijing Key Laboratory of TCVM, Department of Veterinary Medicine, Beijing University of Agriculture, 102206, China
Yan Lu
Beijing Key Laboratory of TCVM, Department of Veterinary Medicine, Beijing University of Agriculture, 102206, China
Yuanyuan Ma
Laboratory Animal Center, Peking University, 100034, China
Guoying Wu
Maternity and Child Care Center, Changchun City, Jilin, 130600, China
Ross C. Beier
USDA, Agricultural Research Service, Southern Plains Agricultural Research Center, Food and Feed Safety Research Unit, College Station, Texas, USA
Xiaolin Hou
Beijing Key Laboratory of TCVM, Department of Veterinary Medicine, Beijing University of Agriculture, 102206, China
Guojuan Wu
Beijing Key Laboratory of TCVM, Department of Veterinary Medicine, Beijing University of Agriculture, 102206, China