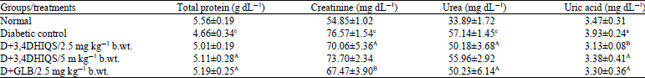
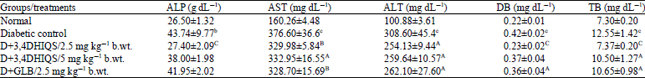
Research Article
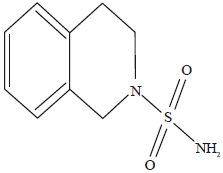
Antidiabetic and Hypolipidemic Potential of 3, 4-dihydroisoquinolin-2(1H)- Sulfonamide in Alloxan Induced Diabetic Rats
Laboratory of Biochemistry and Applied Microbiology, Department of Biochemistry, Faculty of Sciences, University of Badji Mokhtar-Annaba, Algeria
Y. Reggami
Laboratory of Biochemistry and Applied Microbiology, Department of Biochemistry, Faculty of Sciences, University of Badji Mokhtar-Annaba, Algeria
M. Benlaifa
Laboratory of Cellular Toxicology, Department of Biology, Faculty of Sciences, University of Badji Mokhtar-Annaba, Algeria
M. Berredjem
Laboratory of Applied Organic Chemistry, Synthesis of Biomolecules and Molecular Modelling Group, Department of Chemistry, University of Badji Mokhtar-Annaba, Algeria
N. Bouzerna
Laboratory of Biochemistry and Applied Microbiology, Department of Biochemistry, Faculty of Sciences, University of Badji Mokhtar-Annaba, Algeria