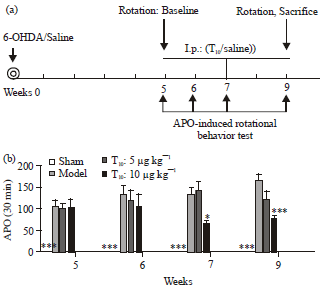
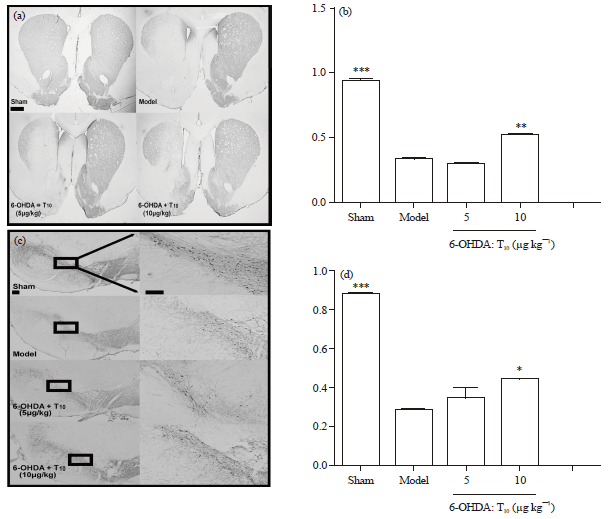
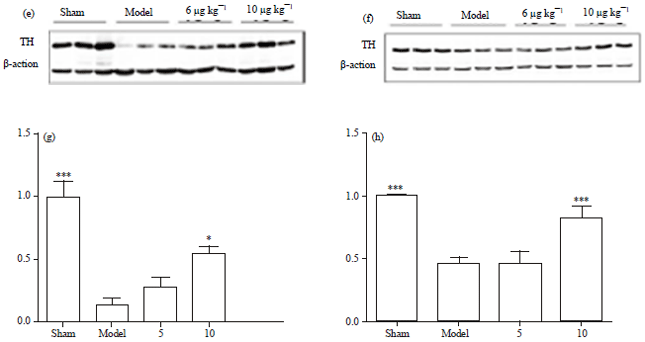
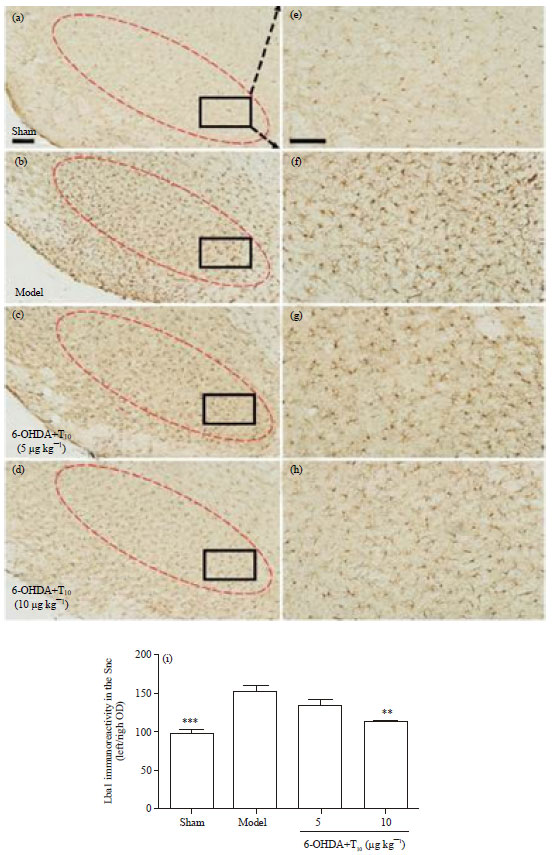
Research Article
Triptolide Protects Dopaminergic Neurons from 6-OHDA Lesion in a Rat Model of Parkinson’s Disease
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Haiting An
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Feilong Zhang
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Lin Dong
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Qi Wang
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Ruijun Su
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Yanjing Qian
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China
Xiaoli Gong
Department of Neurobiology, Key Laboratory for Neurodegenerative Disorder of the Ministry of Education, Beijing Institute of Brain Disorders, Capital Medical University, 10 Xitoutiao, Youanmen, Beijing 100069, People�s Republic of China