Research Article
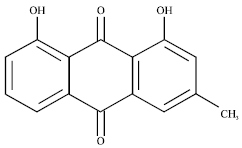
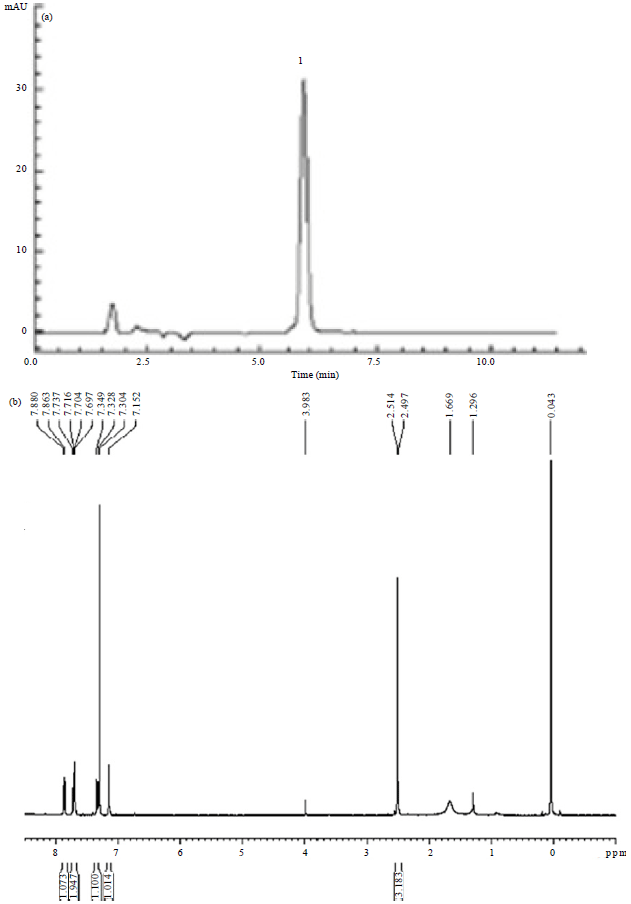
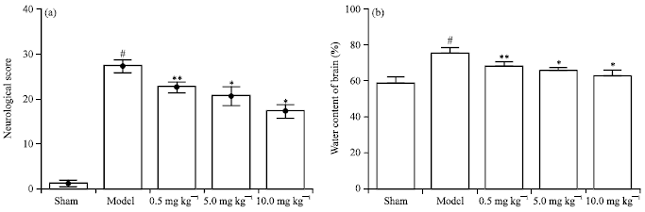
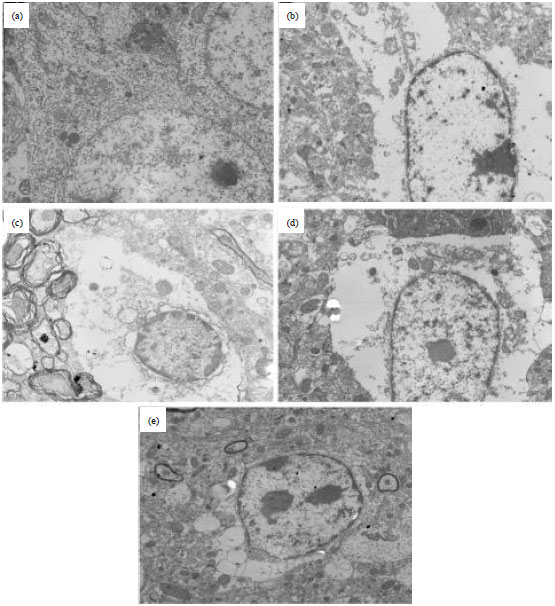
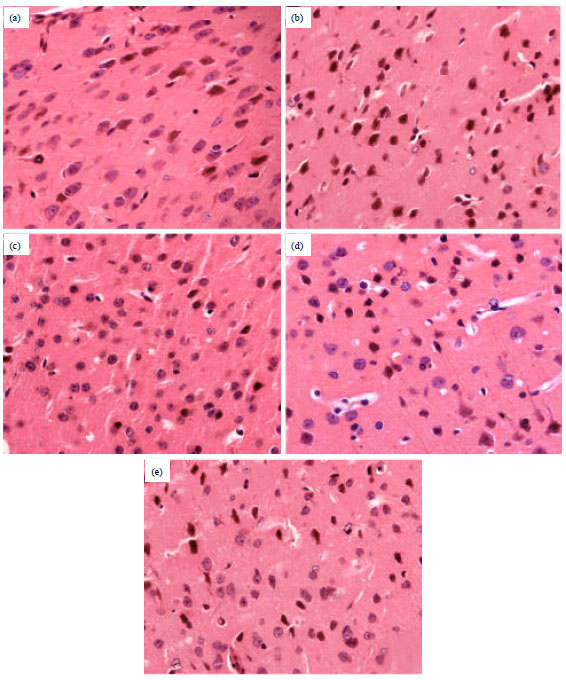
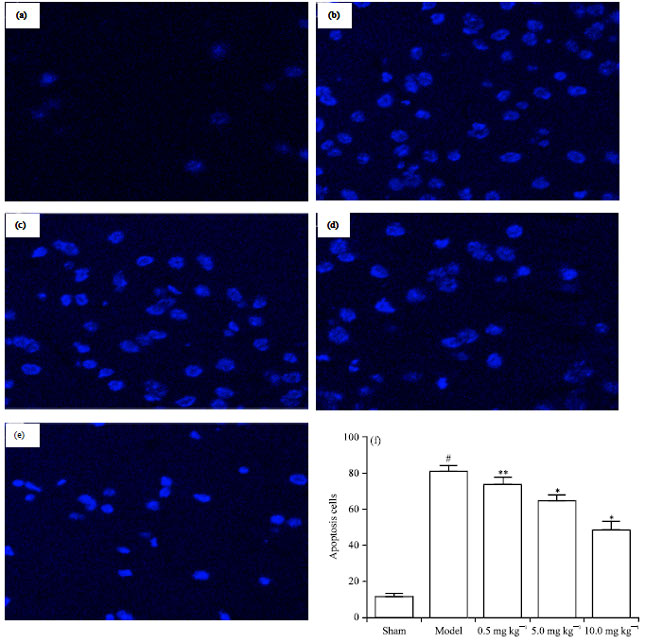
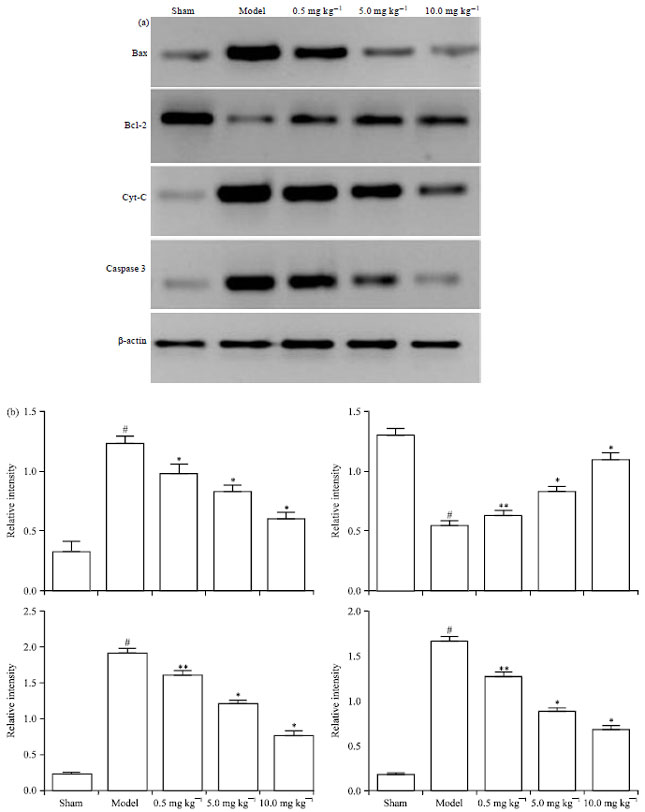
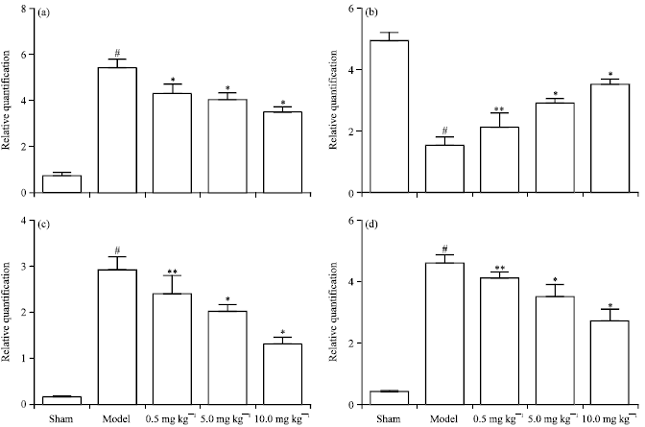
Chrysophanol Liposome Preconditioning Protects against Cerebral Ischemia-reperfusion Injury by Inhibiting Oxidative Stress and Apoptosis in Mice
Department of Pharmacology, Hebei Medical University, Shijiazhuang, People`s Republic of China
Maodong Zheng
Department of Pharmacy, The First Affiliated Hospital of Hebei North University, Zhang Jiakou, People`s Republic of China
Danshen Zhang
Hebei University of Science and Technology, Shijiazhuang, People`s Republic of China