Research Article
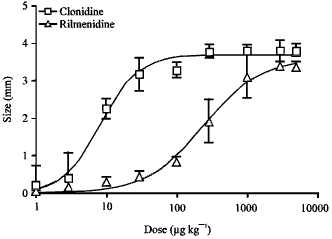
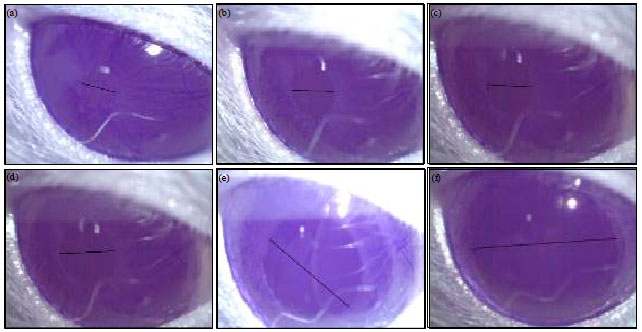
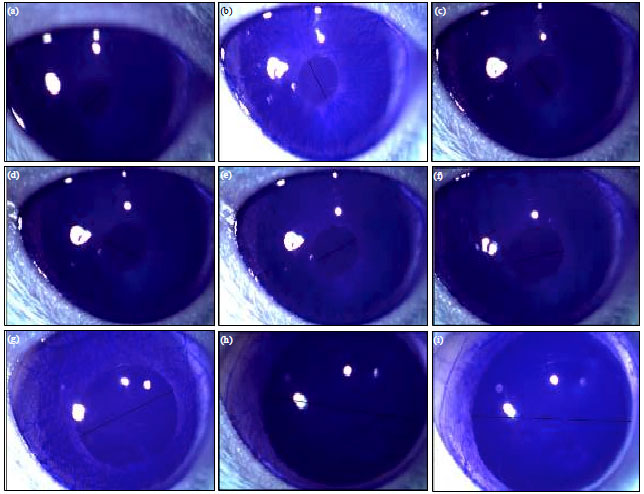
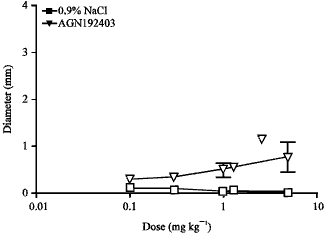
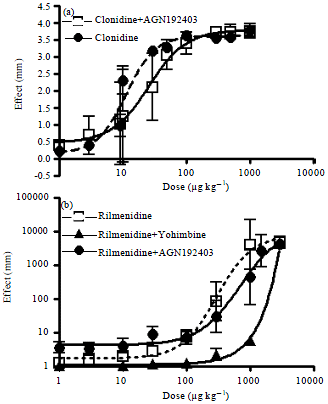
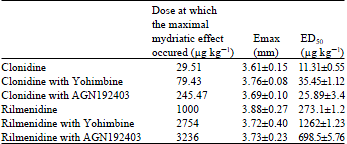
Effect of the Reference Imidazoline Drugs, Clonidine and Rilmenidine, on Rat Eye Pupil Size Confirms the Decisive Role of α2-Adrenoceptors on Mydriasis
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Teresa Frackowiak
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Antoni Nasal
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Joanna Rietz
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Maja Paszek
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Magdalena Buszewska-Forajta
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland
Roman Kaliszan
Department of Biopharmaceutics and Pharmacodynamics, Medical University of Gdask, Gdask, Poland