Research Article
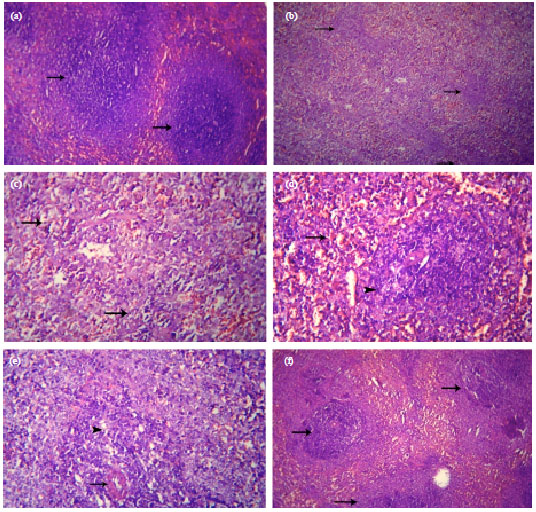
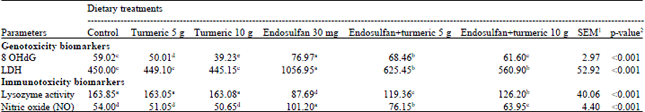
Antidotal Effect of Turmeric (Curcuma longa) against Endosulfan-Induced Cytogenotoxicity and Immunotoxicity in Broiler Chicks
Department of Forensic Medicine and Toxicology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, 44111, Egypt
Mahmoud M. Alagawany
Department of Poultry, Faculty of Agriculture, Zagazig University, Zagazig, 44111, Egypt
Kuldeep Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly, 243122, Uttar Pradesh, India