Research Article
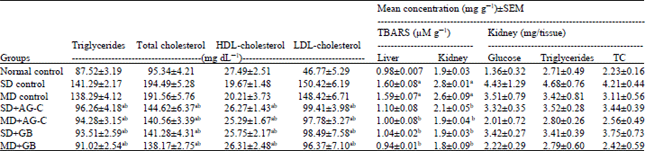
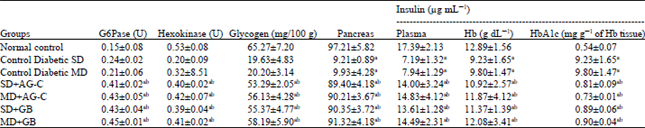
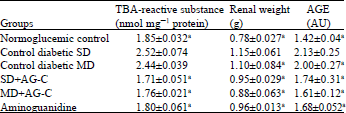
In vitro and in vivo Antidiabetic and Antiglycation Properties of Apium graveolens in Type 1 and 2 Diabetic Rat
Laboratorio de Investigaci�n de Productos Naturales, Escuela Superior de Ingenieria Quimica e Industrias Extractivas IPN. Av. Instituto Politecnico Nacional S/N, Unidad Profesional Adolfo Lopez Mateos CP 07708, Mexico DF
Vicente Aguiniga Juarez
Laboratorio de Investigaci�n de Productos Naturales, Escuela Superior de Ingenieria Quimica e Industrias Extractivas IPN. Av. Instituto Politecnico Nacional S/N, Unidad Profesional Adolfo Lopez Mateos CP 07708, Mexico DF
Jahel Valdes Sauceda
Laboratorio de Investigaci�n de Productos Naturales, Escuela Superior de Ingenieria Quimica e Industrias Extractivas IPN. Av. Instituto Politecnico Nacional S/N, Unidad Profesional Adolfo Lopez Mateos CP 07708, Mexico DF
Irasema Anaya Sosa
Departamento de Graduados de Alimentos, Escuela Nacional de Ciencias Biologicas IPN. Carpio/Plan de Ayala S/N, Santo Tomas Mexico DF