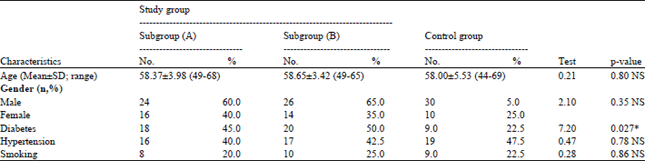
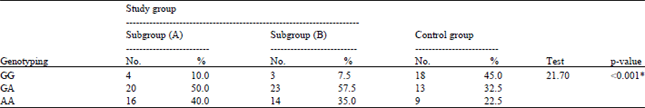
Research Article
Polymorphism of Insulin-Like Growth Factor-II Gene in Primary Open Angle Glaucoma and its Effect on Treatment
Department of Ophthalmology, Benha Faculty of Medicine, Egypt
Omminea Alsaied Abdullah
Department of Biochemistry, Benha Faculty of Medicine, Egypt
Amer Mohammed Abdulhamid
Department of Biochemistry, Al-Azhar Faculty of Medicine, Egypt