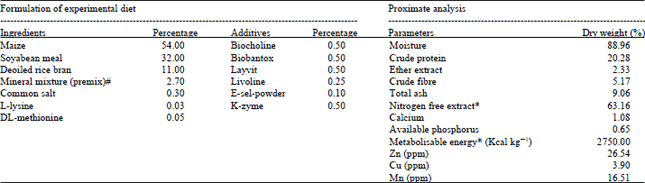
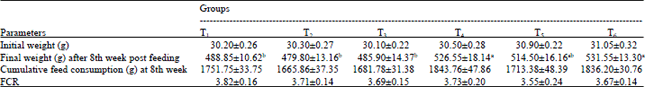
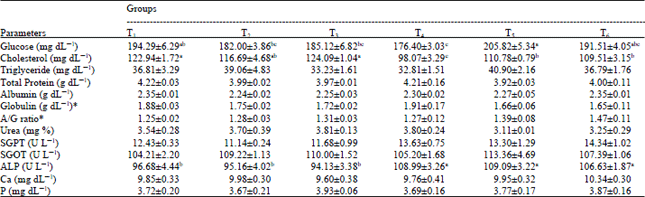
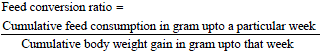Research Article
Effects of Organic Minerals Supplementation on Growth, Bioavailability and Immunity in Layer Chicks
Department of Animal Nutrition, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
S.K. Mishra
Department of Animal Nutrition, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
R.K. Swain
Department of Animal Nutrition, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
G. Sahoo
Department of Veterinary Biochemistry, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
N.C. Behura
Post Graduate Department of Poultry Science, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
K. Sethi
Department of Animal Nutrition, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
B. Chichilichi
Department of Livestock Production Management, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
S.R. Mishra
Department of Animal Nutrition, College of Veterinary Science and Animal Husbandry, Orissa University of Agriculture and Technology, Bhubaneswar, Odisha, 751003, India
T. Behera
Fish Health Management Division, Central Institute of Freshwater Aquaculture, Bhubaneswar, Odisha, 751002, India
K. Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (Uttar Pradesh), 243122, India
P. Swain
Fish Health Management Division, Central Institute of Freshwater Aquaculture, Bhubaneswar, Odisha, 751002, India