Research Article
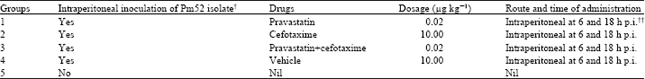
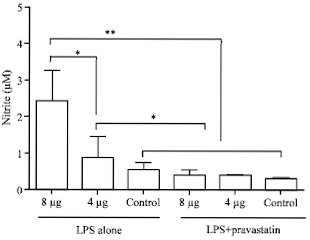
Moderation of Immunopathological Parameters by Pravastatin in Pasteurella multocida (Pm52) Induced Septicaemic Mice
Immunology Section, Izatnagar, Bareilly, Uttar Pradesh, 243122, India
Tapas Kumar Goswami
Immunology Section, Izatnagar, Bareilly, Uttar Pradesh, 243122, India
Raies Ahmad Mir
Division of Bacteriology and Mycology, Izatnagar, Bareilly, Uttar Pradesh, 243122, India
Pallab Chaudhuri
Division of Bacteriology and Mycology, Izatnagar, Bareilly, Uttar Pradesh, 243122, India
Kuldeep Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly, Uttar Pradesh, 243122, India