Research Article
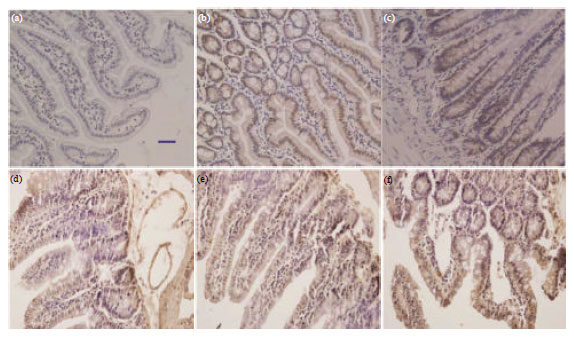
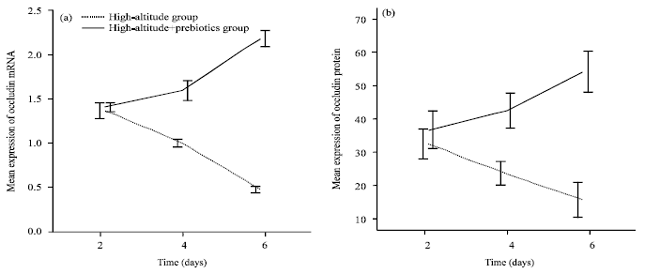
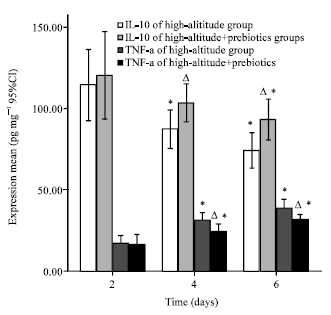
Effects of Prebiotics on the Expression of the Integral Membrane Protein Occludin and Cytokines in Rats after Acute High Altitude Exposure
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Zhi-Yun Deng
Department of Test, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Ju-Zi Dong
Department of Test, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Yun Duan
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Shang-Xin Deng
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Jia-Yu Chen
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Qiang Ma
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China
Ti-Dong Shan
Department of Gastroenterology, General Hospital of Chinese PLA, Lanzhou Command, Lanzhou, 730050, China