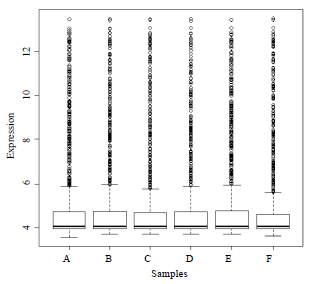
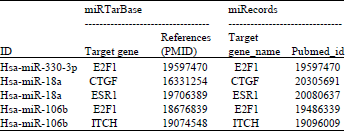
Research Article
MicroRNA Microarray Analysis Combined with Interaction Network Analysis to Investigate the Influence of Clozapine to Metabolic Syndrome
Key Laboratory for the Genetics of Developmental and Neuropsychiatric Disorders, Bio-X Institutes (Ministry of Education), Shanghai Jiao Tong University, 200030, Shanghai, People=s Republic of China
Lin Yang
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Dengtang Liu
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Donghong Cui
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Shunying Yu
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Yan Li
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Haisu Wu
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China
Ying Yue
Shanghai Luwan Mental Health Center, 1162 Quxi Road, 200023, Shanghai, People`s Republic of China
Yongyong Shi
Institute of Neuropsychiatric Science and Systems Biological Medicine, Shanghai Jiao Tong University, 200042, Shanghai, People`s Republic of China
Yifeng Xu
Schizophrenia Program, Shanghai Mental Health Center, School of Medicine, Shanghai Jiao Tong University, 200030 Shanghai, China